Exosomal miR-10a derived from amniotic fluid stem cells preserves ovarian follicles after chemotherapy
- PMID: 26979400
- PMCID: PMC4793229
- DOI: 10.1038/srep23120
Exosomal miR-10a derived from amniotic fluid stem cells preserves ovarian follicles after chemotherapy
Abstract
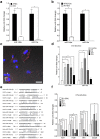
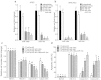
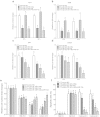
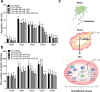
Chemotherapy (CTx)-induced premature ovarian failure (POF) in woman remains clinically irreversible. Amniotic fluid stem cells (AFSCs) have shown the potential to treat CTx-induced POF; however, the underlying mechanism is unclear. Here we demonstrate that AFSC-derived exosomes recapitulate the anti-apoptotic effect of AFSCs on CTx-damaged granulosa cells (GCs), which are vital for the growth of ovarian follicles. AFSC-derived exosomes prevent ovarian follicular atresia in CTx-treated mice via the delivery of microRNAs in which both miR-146a and miR-10a are highly enriched and their potential target genes are critical to apoptosis. The down-regulation of these two miRNAs in AFSC-derived exosomes attenuates the anti-apoptotic effect on CTx-damaged GCs in vitro. Further, the administration of these miRNAs recapitulates the effects both in vitro and in vivo, in which miR-10a contributes a dominant influence. Our findings illustrate that miR-10a has potential as a novel therapeutic agent for the treatment of POF.
Figures
Similar articles
-
The Therapeutic Effect of Stem Cells on Chemotherapy-Induced Premature Ovarian Failure.Curr Mol Med. 2021;21(5):376-384. doi: 10.2174/1566524020666200905113907. Curr Mol Med. 2021. PMID: 32888266 Free PMC article. Review.
-
Human urine stem cells protect against cyclophosphamide-induced premature ovarian failure by inhibiting SLC1A4-mediated outflux of intracellular serine in ovarian granulosa cells.Cell Mol Biol Lett. 2025 Feb 19;30(1):21. doi: 10.1186/s11658-025-00701-1. Cell Mol Biol Lett. 2025. PMID: 39972244 Free PMC article.
-
Amniotic fluid stem cells prevent follicle atresia and rescue fertility of mice with premature ovarian failure induced by chemotherapy.PLoS One. 2014 Sep 8;9(9):e106538. doi: 10.1371/journal.pone.0106538. eCollection 2014. PLoS One. 2014. PMID: 25198549 Free PMC article.
-
Bone marrow mesenchymal stem cell-derived exosomal miR-144-5p improves rat ovarian function after chemotherapy-induced ovarian failure by targeting PTEN.Lab Invest. 2020 Mar;100(3):342-352. doi: 10.1038/s41374-019-0321-y. Epub 2019 Sep 19. Lab Invest. 2020. PMID: 31537899
-
Protective Effects of Antioxidants on Cyclophosphamide-Induced Ovarian Toxicity.Biopreserv Biobank. 2023 Apr;21(2):121-141. doi: 10.1089/bio.2021.0159. Epub 2022 Jun 13. Biopreserv Biobank. 2023. PMID: 35696235 Review.
Cited by
-
The Therapeutic Effect of Stem Cells on Chemotherapy-Induced Premature Ovarian Failure.Curr Mol Med. 2021;21(5):376-384. doi: 10.2174/1566524020666200905113907. Curr Mol Med. 2021. PMID: 32888266 Free PMC article. Review.
-
Research progress of extracellular vesicles in the treatment of ovarian diseases (Review).Exp Ther Med. 2023 Nov 15;27(1):15. doi: 10.3892/etm.2023.12303. eCollection 2024 Jan. Exp Ther Med. 2023. PMID: 38125352 Free PMC article. Review.
-
MicroRNA Shuttle from Cell-To-Cell by Exosomes and Its Impact in Cancer.Noncoding RNA. 2019 Mar 21;5(1):28. doi: 10.3390/ncrna5010028. Noncoding RNA. 2019. PMID: 30901915 Free PMC article. Review.
-
Frontier Review of the Molecular Mechanisms and Current Approaches of Stem Cell-Derived Exosomes.Cells. 2023 Mar 26;12(7):1018. doi: 10.3390/cells12071018. Cells. 2023. PMID: 37048091 Free PMC article. Review.
-
Therapeutic efficacy and promise of stem cell-derived extracellular vesicles in Alzheimer's disease and other aging-related disorders.Ageing Res Rev. 2023 Dec;92:102088. doi: 10.1016/j.arr.2023.102088. Epub 2023 Oct 11. Ageing Res Rev. 2023. PMID: 37827304 Free PMC article. Review.
References
-
- Helleday T., Petermann E., Lundin C., Hodgson B. & Sharma R. A. DNA repair pathways as targets for cancer therapy. Nature Reviews Cancer 8, 193–204 (2008). - PubMed
-
- Zhao X. J., Huang Y. H., Yu Y. C. & Xin X. Y. GnRH antagonist cetrorelix inhibits mitochondria-dependent apoptosis triggered by chemotherapy in granulosa cells of rats. Gynecol. Oncol. 118, 69–75 (2010). - PubMed
-
- Jemal A. et al. Cancer statistics, 2007. CA Cancer J. Clin. 57, 43–66 (2007). - PubMed
-
- Desmeules P. & Devine P. J. Characterizing the ovotoxicity of cyclophosphamide metabolites on cultured mouse ovaries. Toxicol. Sci. 90, 500–509 (2006). - PubMed
-
- Matzuk M. M., Burns K. H., Viveiros M. M. & Eppig J. J. Intercellular communication in the mammalian ovary: oocytes carry the conversation. Science 296, 2178–2180 (2002). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

