Monocarboxylate transporters in the brain and in cancer
- PMID: 26993058
- PMCID: PMC4990061
- DOI: 10.1016/j.bbamcr.2016.03.013
Monocarboxylate transporters in the brain and in cancer
Abstract
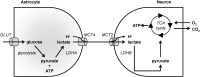
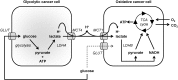
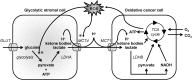
Monocarboxylate transporters (MCTs) constitute a family of 14 members among which MCT1-4 facilitate the passive transport of monocarboxylates such as lactate, pyruvate and ketone bodies together with protons across cell membranes. Their anchorage and activity at the plasma membrane requires interaction with chaperon protein such as basigin/CD147 and embigin/gp70. MCT1-4 are expressed in different tissues where they play important roles in physiological and pathological processes. This review focuses on the brain and on cancer. In the brain, MCTs control the delivery of lactate, produced by astrocytes, to neurons, where it is used as an oxidative fuel. Consequently, MCT dysfunctions are associated with pathologies of the central nervous system encompassing neurodegeneration and cognitive defects, epilepsy and metabolic disorders. In tumors, MCTs control the exchange of lactate and other monocarboxylates between glycolytic and oxidative cancer cells, between stromal and cancer cells and between glycolytic cells and endothelial cells. Lactate is not only a metabolic waste for glycolytic cells and a metabolic fuel for oxidative cells, but it also behaves as a signaling agent that promotes angiogenesis and as an immunosuppressive metabolite. Because MCTs gate the activities of lactate, drugs targeting these transporters have been developed that could constitute new anticancer treatments. This article is part of a Special Issue entitled: Mitochondrial Channels edited by Pierre Sonveaux, Pierre Maechler and Jean-Claude Martinou.
Keywords: Astrocytes; Lactate shuttle; Metabolic cooperation; Neurons; Tumor cells; Tumor microenvironment.
Copyright © 2016 The Authors. Published by Elsevier B.V. All rights reserved.
Figures


References
-
- Halestrap A.P., Meredith D. The SLC16 gene family-from monocarboxylate transporters (MCTs) to aromatic amino acid transporters and beyond. Pflugers Arch. 2004;447:619–628. - PubMed
-
- Murakami Y., Kohyama N., Kobayashi Y., Ohbayashi M., Ohtani H., Sawada Y., Yamamoto T. Functional characterization of human monocarboxylate transporter 6 (SLC16A5) Drug Metab. Dispos. 2005;33:1845–1851. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

