Oxidative stress in retinal pigment epithelium cells increases exosome secretion and promotes angiogenesis in endothelial cells
- PMID: 26999719
- PMCID: PMC4956947
- DOI: 10.1111/jcmm.12834
Oxidative stress in retinal pigment epithelium cells increases exosome secretion and promotes angiogenesis in endothelial cells
Abstract
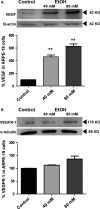
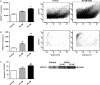
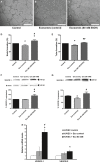
The retinal pigment epithelium (RPE), a monolayer located between the photoreceptors and the choroid, is constantly damaged by oxidative stress, particularly because of reactive oxygen species (ROS). As the RPE, because of its physiological functions, is essential for the survival of the retina, any sustained damage may consequently lead to loss of vision. Exosomes are small membranous vesicles released into the extracellular medium by numerous cell types, including RPE cells. Their cargo includes genetic material and proteins, making these vesicles essential for cell-to-cell communication. Exosomes may fuse with neighbouring cells influencing their fate. It has been observed that RPE cells release higher amounts of exosomes when they are under oxidative stress. Exosomes derived from cultured RPE cells were isolated by ultracentrifugation and quantified by flow cytometry. VEGF receptors (VEGFR) were analysed by both flow cytometry and Western blot. RT-PCR and qPCR were conducted to assess mRNA content of VEGFRs in exosomes. Neovascularization assays were performed after applying RPE exosomes into endothelial cell cultures. Our results showed that stressed RPE cells released a higher amount of exosomes than controls, with a higher expression of VEGFR in the membrane, and enclosed an extra cargo of VEGFR mRNA. Angiogenesis assays confirmed that endothelial cells increased their tube formation capacity when exposed to stressed RPE exosomes.
Keywords: VEGF receptors; angiogenesis; exosomes; oxidative stress; retinal pigment epithelium.
© 2016 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures


Similar articles
-
ARPE-19-derived VEGF-containing exosomes promote neovascularization in HUVEC: the role of the melanocortin receptor 5.Cell Cycle. 2019 Feb;18(4):413-424. doi: 10.1080/15384101.2019.1568745. Epub 2019 Feb 9. Cell Cycle. 2019. PMID: 30739530 Free PMC article.
-
Aliskiren decreases oxidative stress and angiogenic markers in retinal pigment epithelium cells.Angiogenesis. 2017 Feb;20(1):175-181. doi: 10.1007/s10456-016-9526-5. Epub 2016 Oct 1. Angiogenesis. 2017. PMID: 27695972
-
Effects of p75 neurotrophin receptor on regulating hypoxia-induced angiogenic factors in retinal pigment epithelial cells.Mol Cell Biochem. 2015 Jan;398(1-2):123-34. doi: 10.1007/s11010-014-2212-2. Epub 2014 Sep 9. Mol Cell Biochem. 2015. PMID: 25200140
-
Desmosome and Hemidesmosome Disassembly in Retinal Pigmented Epithelium: Intersection with the Exosome Pathway.Adv Exp Med Biol. 2025;1468:339-343. doi: 10.1007/978-3-031-76550-6_56. Adv Exp Med Biol. 2025. PMID: 39930219 Review.
-
Controlled exosome release from the retinal pigment epithelium in situ.Exp Eye Res. 2014 Dec;129:1-4. doi: 10.1016/j.exer.2014.10.010. Epub 2014 Oct 11. Exp Eye Res. 2014. PMID: 25311167 Review.
Cited by
-
Ovarian aging increases small extracellular vesicle CD81+ release in human follicular fluid and influences miRNA profiles.Aging (Albany NY). 2020 Jun 17;12(12):12324-12341. doi: 10.18632/aging.103441. Epub 2020 Jun 17. Aging (Albany NY). 2020. PMID: 32554857 Free PMC article.
-
Exosomes: Insights from Retinoblastoma and Other Eye Cancers.Int J Mol Sci. 2020 Sep 25;21(19):7055. doi: 10.3390/ijms21197055. Int J Mol Sci. 2020. PMID: 32992741 Free PMC article. Review.
-
Crosstalk between Oxidative Stress and Exosomes.Oxid Med Cell Longev. 2022 Aug 30;2022:3553617. doi: 10.1155/2022/3553617. eCollection 2022. Oxid Med Cell Longev. 2022. PMID: 36082080 Free PMC article. Review.
-
Extracellular vesicles regulate the transmission of insulin resistance and redefine noncommunicable diseases.Front Mol Biosci. 2023 Jan 9;9:1024786. doi: 10.3389/fmolb.2022.1024786. eCollection 2022. Front Mol Biosci. 2023. PMID: 36699697 Free PMC article. Review.
-
Endogenous and Exogenous Regulation of Redox Homeostasis in Retinal Pigment Epithelium Cells: An Updated Antioxidant Perspective.Int J Mol Sci. 2023 Jun 28;24(13):10776. doi: 10.3390/ijms241310776. Int J Mol Sci. 2023. PMID: 37445953 Free PMC article. Review.
References
-
- Van Deun J, Mestdagh P, Sormunen R, et al The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling. J Extracell Vesicles. 2014; 3: doi:10.3402/jev.v3.24858. - DOI - PMC - PubMed
-
- Geminard C, Nault F, Johnstone RM, et al Characteristics of the interaction between Hsc70 and the transferring receptor in exosomes released during reticulocyte maturation. J Biol Chem. 2001; 276: 9910–6. - PubMed
-
- Janowska‐Wieczorek A, Wysoczynski M, Kijowski J, et al Microvesicles derived from activated platelets induce metastasis and angiogenesis in lung cancer. Int J Cancer. 2005; 113: 752–60. - PubMed
-
- Taieb J, Chaput N, Zitvogel L. Dendritic cellderived exosomes as cell‐free peptide‐based vaccines. Crit Rev Immunol. 2005; 25: 215–23. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

