Propionate Increases Hepatic Pyruvate Cycling and Anaplerosis and Alters Mitochondrial Metabolism
- PMID: 27002151
- PMCID: PMC4933266
- DOI: 10.1074/jbc.M116.720631
Propionate Increases Hepatic Pyruvate Cycling and Anaplerosis and Alters Mitochondrial Metabolism
Abstract
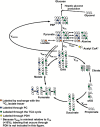
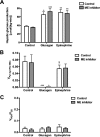
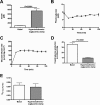
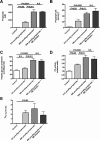
In mammals, pyruvate kinase (PK) plays a key role in regulating the balance between glycolysis and gluconeogenesis; however, in vivo regulation of PK flux by gluconeogenic hormones and substrates is poorly understood. To this end, we developed a novel NMR-liquid chromatography/tandem-mass spectrometry (LC-MS/MS) method to directly assess pyruvate cycling relative to mitochondrial pyruvate metabolism (VPyr-Cyc/VMito) in vivo using [3-(13)C]lactate as a tracer. Using this approach, VPyr-Cyc/VMito was only 6% in overnight fasted rats. In contrast, when propionate was infused simultaneously at doses previously used as a tracer, it increased VPyr-Cyc/VMito by 20-30-fold, increased hepatic TCA metabolite concentrations 2-3-fold, and increased endogenous glucose production rates by 20-100%. The physiologic stimuli, glucagon and epinephrine, both increased hepatic glucose production, but only glucagon suppressed VPyr-Cyc/VMito These data show that under fasting conditions, when hepatic gluconeogenesis is stimulated, pyruvate recycling is relatively low in liver compared with VMito flux and that liver metabolism, in particular pyruvate cycling, is sensitive to propionate making it an unsuitable tracer to assess hepatic glycolytic, gluconeogenic, and mitochondrial metabolism in vivo.
Keywords: epinephrine; glucagon; gluconeogenesis; isotopic tracer; liver metabolism; pyruvate kinase.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


References
-
- Kumashiro N., Beddow S. A., Vatner D. F., Majumdar S. K., Cantley J. L., Guebre-Egziabher F., Fat I., Guigni B., Jurczak M. J., Birkenfeld A. L., Kahn M., Perler B. K., Puchowicz M. A., Manchem V. P., Bhanot S., et al. (2013) Targeting pyruvate carboxylase reduces gluconeogenesis and adiposity and improves insulin resistance. Diabetes 62, 2183–2194 - PMC - PubMed
-
- Jin E. S., Burgess S. C., Merritt M. E., Sherry A. D., and Malloy C. R. (2005) Differing mechanisms of hepatic glucose overproduction in triiodothyronine-treated rats vs. Zucker diabetic fatty rats by NMR analysis of plasma glucose. Am. J. Physiol. Endocrinol. Metab. 288, E654–E662 - PubMed
-
- Jin E. S., Jones J. G., Merritt M., Burgess S. C., Malloy C. R., and Sherry A. D. (2004) Glucose production, gluconeogenesis, and hepatic tricarboxylic acid cycle fluxes measured by nuclear magnetic resonance analysis of a single glucose derivative. Anal. Biochem. 327, 149–155 - PubMed
-
- Satapati S., Kucejova B., Duarte J. A., Fletcher J. A., Reynolds L., Sunny N. E., He T., Nair L. A., Livingston K., Fu X., Merritt M. E., Sherry A. D., Malloy C. R., Shelton J. M., Lambert J., et al. (2015) Mitochondrial metabolism mediates oxidative stress and inflammation in fatty liver. J. Clin. Invest. 125, 4447–4462 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

