Recycling Endosomes and Viral Infection
- PMID: 27005655
- PMCID: PMC4810254
- DOI: 10.3390/v8030064
Recycling Endosomes and Viral Infection
Abstract
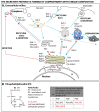
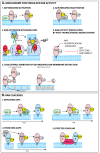
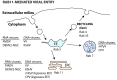
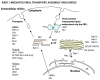
Many viruses exploit specific arms of the endomembrane system. The unique composition of each arm prompts the development of remarkably specific interactions between viruses and sub-organelles. This review focuses on the viral-host interactions occurring on the endocytic recycling compartment (ERC), and mediated by its regulatory Ras-related in brain (Rab) GTPase Rab11. This protein regulates trafficking from the ERC and the trans-Golgi network to the plasma membrane. Such transport comprises intricate networks of proteins/lipids operating sequentially from the membrane of origin up to the cell surface. Rab11 is also emerging as a critical factor in an increasing number of infections by major animal viruses, including pathogens that provoke human disease. Understanding the interplay between the ERC and viruses is a milestone in human health. Rab11 has been associated with several steps of the viral lifecycles by unclear processes that use sophisticated diversified host machinery. For this reason, we first explore the state-of-the-art on processes regulating membrane composition and trafficking. Subsequently, this review outlines viral interactions with the ERC, highlighting current knowledge on viral-host binding partners. Finally, using examples from the few mechanistic studies available we emphasize how ERC functions are adjusted during infection to remodel cytoskeleton dynamics, innate immunity and membrane composition.
Keywords: Rab GTPases; Rab11; animal viruses; intracellular membranes; phosphoinositides; recycling endosome.
Figures


Similar articles
-
Budding of Ebola Virus Particles Requires the Rab11-Dependent Endocytic Recycling Pathway.J Infect Dis. 2018 Nov 22;218(suppl_5):S388-S396. doi: 10.1093/infdis/jiy460. J Infect Dis. 2018. PMID: 30476249 Free PMC article.
-
Orchestration of cell surface proteins by Rab11.Trends Cell Biol. 2014 Jul;24(7):407-15. doi: 10.1016/j.tcb.2014.02.004. Epub 2014 Mar 24. Trends Cell Biol. 2014. PMID: 24675420 Review.
-
Evidence for the role of Rab11-positive recycling endosomes as intermediates in coronavirus egress from epithelial cells.Histochem Cell Biol. 2022 Sep;158(3):241-251. doi: 10.1007/s00418-022-02115-y. Epub 2022 May 23. Histochem Cell Biol. 2022. PMID: 35604431 Free PMC article.
-
Rab11 activity and PtdIns(3)P turnover removes recycling cargo from endosomes.Nat Chem Biol. 2018 Aug;14(8):801-810. doi: 10.1038/s41589-018-0086-4. Epub 2018 Jun 18. Nat Chem Biol. 2018. PMID: 29915378
-
Viral interactions with host cell Rab GTPases.Small GTPases. 2018 Mar 4;9(1-2):192-201. doi: 10.1080/21541248.2017.1346552. Epub 2017 Sep 18. Small GTPases. 2018. PMID: 28696820 Free PMC article. Review.
Cited by
-
ICAM-1 Binding Rhinoviruses A89 and B14 Uncoat in Different Endosomal Compartments.J Virol. 2016 Aug 12;90(17):7934-42. doi: 10.1128/JVI.00712-16. Print 2016 Sep 1. J Virol. 2016. PMID: 27334586 Free PMC article.
-
Cytomegaloviruses Exploit Recycling Rab Proteins in the Sequential Establishment of the Assembly Compartment.Front Cell Dev Biol. 2018 Dec 4;6:165. doi: 10.3389/fcell.2018.00165. eCollection 2018. Front Cell Dev Biol. 2018. PMID: 30564576 Free PMC article. Review.
-
The Endosomal Recycling Pathway-At the Crossroads of the Cell.Int J Mol Sci. 2020 Aug 23;21(17):6074. doi: 10.3390/ijms21176074. Int J Mol Sci. 2020. PMID: 32842549 Free PMC article. Review.
-
Budding of Ebola Virus Particles Requires the Rab11-Dependent Endocytic Recycling Pathway.J Infect Dis. 2018 Nov 22;218(suppl_5):S388-S396. doi: 10.1093/infdis/jiy460. J Infect Dis. 2018. PMID: 30476249 Free PMC article.
-
Allosteric binding sites in Rab11 for potential drug candidates.PLoS One. 2018 Jun 6;13(6):e0198632. doi: 10.1371/journal.pone.0198632. eCollection 2018. PLoS One. 2018. PMID: 29874286 Free PMC article.
References
-
- Robbins F.C., Enders J.F., Weller T.H. The effect of poliomyelitis virus upon cells in tissue cultures. J. Clin. Investig. 1950;29:841. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

