Transplantation of Nonexpanded Adipose Stromal Vascular Fraction and Platelet-Rich Plasma for Articular Cartilage Injury Treatment in Mice Model
- PMID: 27006923
- PMCID: PMC4782730
- DOI: 10.1155/2013/832396
Transplantation of Nonexpanded Adipose Stromal Vascular Fraction and Platelet-Rich Plasma for Articular Cartilage Injury Treatment in Mice Model
Abstract
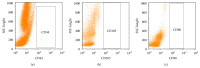
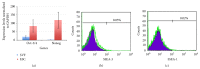
Stromal vascular fraction (SVF) combined with platelet-rich plasma (PRP) is commonly used in preclinical and clinical osteoarthritis as well as articular cartilage injury treatment. However, this therapy has not carefully evaluated the safety and the efficacy. This research aims to assess the safety and the efficacy of SVF combined with PRP transplantation. Ten samples of SVFs and PRPs from donors were used in this research. About safety, we evaluate the expression of some genes related to tumor formation such as Oct-4, Nanog, SSEA3, and SSEA4 by RT-PCR, flow cytometry, and tumor formation when injected in NOD/SCID mice. About efficacy, SVF was injected with PRP into murine joint that caused joint failure. The results showed that SVFs are negative with Oct-4, Nanog, SSEA-3, and SSEA-4, as well as they cannot cause tumors in mice. SVFs combined with PRP can improve the joint regeneration in mice. These results proved that SVFs combined with PRP transplantation is a promising therapy for articular cartilage injury treatment.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
