Thalamic reticular impairment underlies attention deficit in Ptchd1(Y/-) mice
- PMID: 27007844
- PMCID: PMC4875756
- DOI: 10.1038/nature17427
Thalamic reticular impairment underlies attention deficit in Ptchd1(Y/-) mice
Abstract
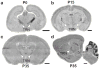
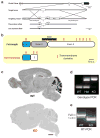
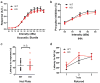
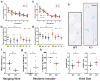
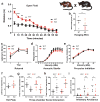
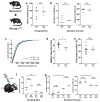
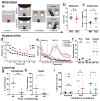
Developmental disabilities, including attention-deficit hyperactivity disorder (ADHD), intellectual disability (ID), and autism spectrum disorders (ASD), affect one in six children in the USA. Recently, gene mutations in patched domain containing 1 (PTCHD1) have been found in ~1% of patients with ID and ASD. Individuals with PTCHD1 deletion show symptoms of ADHD, sleep disruption, hypotonia, aggression, ASD, and ID. Although PTCHD1 is probably critical for normal development, the connection between its deletion and the ensuing behavioural defects is poorly understood. Here we report that during early post-natal development, mouse Ptchd1 is selectively expressed in the thalamic reticular nucleus (TRN), a group of GABAergic neurons that regulate thalamocortical transmission, sleep rhythms, and attention. Ptchd1 deletion attenuates TRN activity through mechanisms involving small conductance calcium-dependent potassium currents (SK). TRN-restricted deletion of Ptchd1 leads to attention deficits and hyperactivity, both of which are rescued by pharmacological augmentation of SK channel activity. Global Ptchd1 deletion recapitulates learning impairment, hyper-aggression, and motor defects, all of which are insensitive to SK pharmacological targeting and not found in the TRN-restricted deletion mouse. This study maps clinically relevant behavioural phenotypes onto TRN dysfunction in a human disease model, while also identifying molecular and circuit targets for intervention.
Conflict of interest statement
The authors declare no competing financial interests.
Figures









Comment in
-
Neuroscience: Untangling autism.Nature. 2016 Apr 7;532(7597):45-6. doi: 10.1038/nature17311. Epub 2016 Mar 23. Nature. 2016. PMID: 27007842 Free PMC article.
References
-
- Cristino AS, et al. Neurodevelopmental and neuropsychiatric disorders represent an interconnected molecular system. Mol Psychiatry. 2014;19(3):294–301. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

