Proximal tubule-specific glutamine synthetase deletion alters basal and acidosis-stimulated ammonia metabolism
- PMID: 27009341
- PMCID: PMC4935770
- DOI: 10.1152/ajprenal.00547.2015
Proximal tubule-specific glutamine synthetase deletion alters basal and acidosis-stimulated ammonia metabolism
Abstract
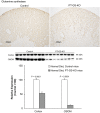
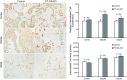
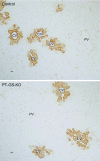
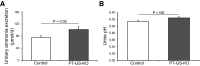
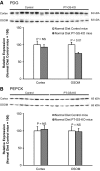
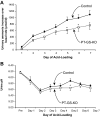
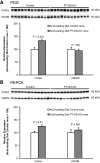
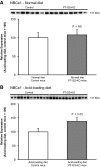
Glutamine synthetase (GS) catalyzes the recycling of NH4 (+) with glutamate to form glutamine. GS is highly expressed in the renal proximal tubule (PT), suggesting ammonia recycling via GS could decrease net ammoniagenesis and thereby limit ammonia available for net acid excretion. The purpose of the present study was to determine the role of PT GS in ammonia metabolism under basal conditions and during metabolic acidosis. We generated mice with PT-specific GS deletion (PT-GS-KO) using Cre-loxP techniques. Under basal conditions, PT-GS-KO increased urinary ammonia excretion significantly. Increased ammonia excretion occurred despite decreased expression of key proteins involved in renal ammonia generation. After the induction of metabolic acidosis, the ability to increase ammonia excretion was impaired significantly by PT-GS-KO. The blunted increase in ammonia excretion occurred despite greater expression of multiple components of ammonia generation, including SN1 (Slc38a3), phosphate-dependent glutaminase, phosphoenolpyruvate carboxykinase, and Na(+)-coupled electrogenic bicarbonate cotransporter. We conclude that 1) GS-mediated ammonia recycling in the PT contributes to both basal and acidosis-stimulated ammonia metabolism and 2) adaptive changes in other proteins involved in ammonia metabolism occur in response to PT-GS-KO and cause an underestimation of the role of PT GS expression.
Keywords: acid-base; ammonia; glutamine synthetase; proximal tubule.
Figures





Similar articles
-
Proximal tubule glutamine synthetase expression is necessary for the normal response to dietary protein restriction.Am J Physiol Renal Physiol. 2017 Jul 1;313(1):F116-F125. doi: 10.1152/ajprenal.00048.2017. Epub 2017 Mar 22. Am J Physiol Renal Physiol. 2017. PMID: 28331060 Free PMC article.
-
NBCe1-A Regulates Proximal Tubule Ammonia Metabolism under Basal Conditions and in Response to Metabolic Acidosis.J Am Soc Nephrol. 2018 Apr;29(4):1182-1197. doi: 10.1681/ASN.2017080935. Epub 2018 Feb 26. J Am Soc Nephrol. 2018. PMID: 29483156 Free PMC article.
-
Acid-base effects of combined renal deletion of NBCe1-A and NBCe1-B.Am J Physiol Renal Physiol. 2022 Feb 1;322(2):F208-F224. doi: 10.1152/ajprenal.00358.2021. Epub 2022 Jan 10. Am J Physiol Renal Physiol. 2022. PMID: 35001662 Free PMC article.
-
Recent advances in understanding renal ammonia metabolism and transport.Curr Opin Nephrol Hypertens. 2016 Sep;25(5):436-43. doi: 10.1097/MNH.0000000000000255. Curr Opin Nephrol Hypertens. 2016. PMID: 27367914 Free PMC article. Review.
-
Emerging Features of Ammonia Metabolism and Transport in Acid-Base Balance.Semin Nephrol. 2019 Jul;39(4):394-405. doi: 10.1016/j.semnephrol.2019.04.008. Semin Nephrol. 2019. PMID: 31300094 Free PMC article. Review.
Cited by
-
Proximal tubule glutamine synthetase expression is necessary for the normal response to dietary protein restriction.Am J Physiol Renal Physiol. 2017 Jul 1;313(1):F116-F125. doi: 10.1152/ajprenal.00048.2017. Epub 2017 Mar 22. Am J Physiol Renal Physiol. 2017. PMID: 28331060 Free PMC article.
-
The proximal tubule through an NBCe1-dependent mechanism regulates collecting duct phenotypic and remodeling responses to acidosis.Am J Physiol Renal Physiol. 2023 Jan 1;324(1):F12-F29. doi: 10.1152/ajprenal.00175.2022. Epub 2022 Oct 20. Am J Physiol Renal Physiol. 2023. PMID: 36264886 Free PMC article.
-
NBCe1-A Regulates Proximal Tubule Ammonia Metabolism under Basal Conditions and in Response to Metabolic Acidosis.J Am Soc Nephrol. 2018 Apr;29(4):1182-1197. doi: 10.1681/ASN.2017080935. Epub 2018 Feb 26. J Am Soc Nephrol. 2018. PMID: 29483156 Free PMC article.
-
Expression of sodium-dependent dicarboxylate transporter 1 (NaDC1/SLC13A2) in normal and neoplastic human kidney.Am J Physiol Renal Physiol. 2017 Mar 1;312(3):F427-F435. doi: 10.1152/ajprenal.00559.2016. Epub 2016 Dec 7. Am J Physiol Renal Physiol. 2017. PMID: 27927654 Free PMC article.
-
Acetazolamide causes renal wasting but inhibits ammoniagenesis and prevents the correction of metabolic acidosis by the kidney.Am J Physiol Renal Physiol. 2020 Sep 1;319(3):F366-F379. doi: 10.1152/ajprenal.00501.2019. Epub 2020 Jul 13. Am J Physiol Renal Physiol. 2020. PMID: 32657159 Free PMC article.
References
-
- Biver S, Belge H, Bourgeois S, Van Vooren P, Nowik M, Scohy S, Houillier P, Szpirer J, Szpirer C, Wagner CA, Devuyst O, Marini AM. A role for Rhesus factor Rhcg in renal ammonium excretion and male fertility. Nature 456: 339–343, 2008. - PubMed
-
- Boulland JL, Osen KK, Levy LM, Danbolt NC, Edwards RH, Storm-Mathisen J, Chaudhry FA. Cell-specific expression of the glutamine transporter SN1 suggests differences in dependence on the glutamine cycle. Eur J Neurosci 15: 1615–1631, 2002. - PubMed
-
- Busque SM, Wagner CA. Potassium restriction, high protein intake, and metabolic acidosis increase expression of the glutamine transporter SNAT3 (Slc38a3) in mouse kidney. Am J Physiol Renal Physiol 297: F440–F450, 2009. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

