Lesion Size Is Exacerbated in Hypoxic Rats Whereas Hypoxia-Inducible Factor-1 Alpha and Vascular Endothelial Growth Factor Increase in Injured Normoxic Rats: A Prospective Cohort Study of Secondary Hypoxia in Focal Traumatic Brain Injury
- PMID: 27014178
- PMCID: PMC4780037
- DOI: 10.3389/fneur.2016.00023
Lesion Size Is Exacerbated in Hypoxic Rats Whereas Hypoxia-Inducible Factor-1 Alpha and Vascular Endothelial Growth Factor Increase in Injured Normoxic Rats: A Prospective Cohort Study of Secondary Hypoxia in Focal Traumatic Brain Injury
Abstract
Background: Hypoxia following traumatic brain injury (TBI) is a severe insult shown to exacerbate the pathophysiology, resulting in worse outcome. The aim of this study was to investigate the effects of a hypoxic insult in a focal TBI model by monitoring brain edema, lesion volume, serum biomarker levels, immune cell infiltration, as well as the expression of hypoxia-inducible factor-1 alpha (HIF-1α) and vascular endothelial growth factor (VEGF).
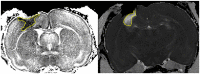
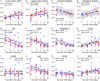
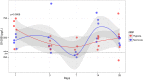
Materials and methods: Female Sprague-Dawley rats (n = 73, including sham and naive) were used. The rats were intubated and mechanically ventilated. A controlled cortical impact device created a 3-mm deep lesion in the right parietal hemisphere. Post-injury, rats inhaled either normoxic (22% O2) or hypoxic (11% O2) mixtures for 30 min. The rats were sacrificed at 1, 3, 7, 14, and 28 days post-injury. Serum was collected for S100B measurements using ELISA. Ex vivo magnetic resonance imaging (MRI) was performed to determine lesion size and edema volume. Immunofluorescence was employed to analyze neuronal death, changes in cerebral macrophage- and neutrophil infiltration, microglia proliferation, apoptosis, complement activation (C5b9), IgG extravasation, HIF-1α, and VEGF.
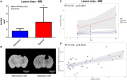
Results: The hypoxic group had significantly increased blood levels of lactate and decreased pO2 (p < 0.0001). On MRI post-traumatic hypoxia resulted in larger lesion areas (p = 0.0173), and NeuN staining revealed greater neuronal loss (p = 0.0253). HIF-1α and VEGF expression was significantly increased in normoxic but not in hypoxic animals (p < 0.05). A trend was seen for serum levels of S100B to be higher in the hypoxic group at 1 day after trauma (p = 0.0868). No differences were observed between the groups in cytotoxic and vascular edema, IgG extravasation, neutrophils and macrophage aggregation, microglia proliferation, or C5b-9 expression.
Conclusion: Hypoxia following focal TBI exacerbated the lesion size and neuronal loss. Moreover, there was a tendency to higher levels of S100B in the hypoxic group early after injury, indicating a potential validity as a biomarker of injury severity. In the normoxic group, the expression of HIF-1α and VEGF was found elevated, possibly indicative of neuro-protective responses occurring in this less severely injured group. Further studies are warranted to better define the pathophysiology of post-TBI hypoxia.
Keywords: S100B; edema; hypoxia; hypoxia-inducible factor 1; neuronal death; secondary insults; traumatic brain injury; vascular endothelial growth factor A.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

