White matter hyperintensities are a core feature of Alzheimer's disease: Evidence from the dominantly inherited Alzheimer network
- PMID: 27016429
- PMCID: PMC4884146
- DOI: 10.1002/ana.24647
White matter hyperintensities are a core feature of Alzheimer's disease: Evidence from the dominantly inherited Alzheimer network
Abstract
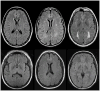
Objective: White matter hyperintensities (WMHs) are areas of increased signal on T2-weighted magnetic resonance imaging (MRI) scans that most commonly reflect small vessel cerebrovascular disease. Increased WMH volume is associated with risk and progression of Alzheimer's disease (AD). These observations are typically interpreted as evidence that vascular abnormalities play an additive, independent role contributing to symptom presentation, but not core features of AD. We examined the severity and distribution of WMH in presymptomatic PSEN1, PSEN2, and APP mutation carriers to determine the extent to which WMH manifest in individuals genetically determined to develop AD.
Methods: The study comprised participants (n = 299; age = 39.03 ± 10.13) from the Dominantly Inherited Alzheimer Network, including 184 (61.5%) with a mutation that results in AD and 115 (38.5%) first-degree relatives who were noncarrier controls. We calculated the estimated years from expected symptom onset (EYO) by subtracting the affected parent's symptom onset age from the participant's age. Baseline MRI data were analyzed for total and regional WMH. Mixed-effects piece-wise linear regression was used to examine WMH differences between carriers and noncarriers with respect to EYO.
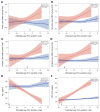
Results: Mutation carriers had greater total WMH volumes, which appeared to increase approximately 6 years before expected symptom onset. Effects were most prominent for the parietal and occipital lobe, which showed divergent effects as early as 22 years before estimated onset.
Interpretation: Autosomal-dominant AD is associated with increased WMH well before expected symptom onset. The findings suggest the possibility that WMHs are a core feature of AD, a potential therapeutic target, and a factor that should be integrated into pathogenic models of the disease. Ann Neurol 2016;79:929-939.
© 2016 American Neurological Association.
Conflict of interest statement
Potential conflicts of interest
SL, FV, MEZ, AN, GT, TLSB, DSM, AF, AG, NJC, DMH, VB, BG, EM, RNM, AJS, CLM, JMR, NR, SF, CL, PRS, SS, SC, CJ, RJB, JCM, RM, and AMB have no relevant conflicts of interest
NF consults for Eli Lilly, Novartis, Sanofi, Roche, and GSK, which may conduct trials in autosomal dominant AD
RAS receives research support from Eli Lilly which manufactures solanezumab, being studied for the treatment of Alzheimer’s disease in the DIAN Study, and from Avid, who manufactures florbetapir, a PET tracer to detect amyloid, that is being used in the DIAN Study.
MW provides consulting to Eli Lilly, which makes an amyloid PET ligand. Eli Lilly is testing anti-amyloid treatments for Alzheimer’s disease.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

