Restimulation After Cryopreservation and Thawing Preserves the Phenotype and Function of Expanded Baboon Regulatory T Cells
- PMID: 27019869
- PMCID: PMC4806341
- DOI: 10.1097/TXD.0000000000000511
Restimulation After Cryopreservation and Thawing Preserves the Phenotype and Function of Expanded Baboon Regulatory T Cells
Abstract
Background: Regulatory T cells (Treg) are being explored for their tolerance-inducing capabilities. Freezing and banking Treg for future use makes this strategy more clinically applicable. We aimed to devise an improved method of expanding and cryopreserving Treg to maximize yield, purity, and function for use in xenotransplantation.
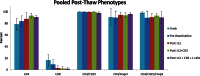
Methods: Baboon peripheral blood mononuclear cells (PBMC) were isolated from whole blood. CD4+/CD25hi cells were isolated by flow cytometric sorting and expanded for 26 days in culture with IL-2, anti-CD3 antibody, artificial APCs transfected with human CD58, CD32, and CD80, and rapamycin with weekly restimulations. Expanded Treg were frozen for 2 months then thawed and cultured for 48 hours in medium plus 1) no additives, 2) IL-2, 3) anti-CD3 antibody, 4) IL-2 + anti-CD3 antibody, and 5) IL-2 + anti-CD3 antibody + L cells. Phenotype and suppression were assessed after expansion, immediately after thawing, and after culturing.
Results: We expanded purified baboon Treg more than 10,000-fold. Expanded Treg exhibited excellent suppression in functional assays. Cryopreservation decreased suppressive function without changing phenotype, but increasing amounts of reactivation after thawing produced significantly better viability and suppressive function with a trend towards greater Treg purity.
Conclusions: We produced numbers of expanded Tregs consistent with clinical use. In contrast to some previous reports, both Treg phenotype and suppressive function were preserved or even enhanced by increasing amounts of restimulation after thawing. Thus, banking of expanded recipient Tregs for in vivo infusion should be possible.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Juvet SC, Whatcott AG, Bushell AR, Wood KJ. Harnessing regulatory T cells for clinical use in transplantation: the end of the beginning. Am J Transplant 2014;14 (4):750. - PubMed
-
- Trzonkowski P, Bieniaszewska M, Juscinska J, et al. First-in-man clinical results of the treatment of patients with graft versus host disease with human ex vivo expanded CD4 + CD25 + CD127− T regulatory cells. Clin Immunol 2009;133 (1):22. - PubMed
-
- Schliesser U, Streitz M, Sawitzki B. Tregs: application for solid-organ transplantation Curr Opin Organ Transplant. 2012;17 (1):34. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

