Modulation of Cortical Oscillations by Low-Frequency Direct Cortical Stimulation Is State-Dependent
- PMID: 27023427
- PMCID: PMC4811434
- DOI: 10.1371/journal.pbio.1002424
Modulation of Cortical Oscillations by Low-Frequency Direct Cortical Stimulation Is State-Dependent
Abstract
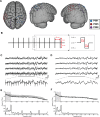
Cortical oscillations play a fundamental role in organizing large-scale functional brain networks. Noninvasive brain stimulation with temporally patterned waveforms such as repetitive transcranial magnetic stimulation (rTMS) and transcranial alternating current stimulation (tACS) have been proposed to modulate these oscillations. Thus, these stimulation modalities represent promising new approaches for the treatment of psychiatric illnesses in which these oscillations are impaired. However, the mechanism by which periodic brain stimulation alters endogenous oscillation dynamics is debated and appears to depend on brain state. Here, we demonstrate with a static model and a neural oscillator model that recurrent excitation in the thalamo-cortical circuit, together with recruitment of cortico-cortical connections, can explain the enhancement of oscillations by brain stimulation as a function of brain state. We then performed concurrent invasive recording and stimulation of the human cortical surface to elucidate the response of cortical oscillations to periodic stimulation and support the findings from the computational models. We found that (1) stimulation enhanced the targeted oscillation power, (2) this enhancement outlasted stimulation, and (3) the effect of stimulation depended on behavioral state. Together, our results show successful target engagement of oscillations by periodic brain stimulation and highlight the role of nonlinear interaction between endogenous network oscillations and stimulation. These mechanistic insights will contribute to the design of adaptive, more targeted stimulation paradigms.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



References
-
- Berger H. Uber das Elektrenkephalogramm des Menschen. Archiv für Psychiatrie und Nervenkrankheiten. 1929;87(1):527–70.
-
- Pfurtscheller G, Stancak A Jr., Neuper C. Event-related synchronization (ERS) in the alpha band—an electrophysiological correlate of cortical idling: a review. International journal of psychophysiology: official journal of the International Organization of Psychophysiology. 1996;24(1–2):39–46. . - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

