A Series of Oleanolic Acid Derivatives as Anti-Hepatitis B Virus Agents: Design, Synthesis, and in Vitro and in Vivo Biological Evaluation
- PMID: 27023498
- PMCID: PMC6273212
- DOI: 10.3390/molecules21040402
A Series of Oleanolic Acid Derivatives as Anti-Hepatitis B Virus Agents: Design, Synthesis, and in Vitro and in Vivo Biological Evaluation
Abstract
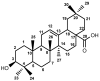
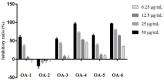
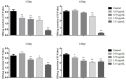
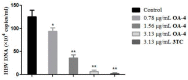
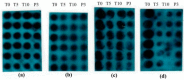
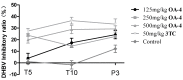
A series of oleanolic acid derivatives were synthesized by diverse reactions, including the introduction of conjugated alkadiene and epoxy ring moieties formed by means of photosensitized oxidation. Eosin Y was used as photosensitizer during this process. Next the cytotoxicity of the products was evaluated on HepG2.2.15 cells to determine the appropriate treatment concentration for the subsequent experiments. Most of the OA derivatives exhibited anti-HBV antigens secretion activity in HepG2.2.15 cells. Among the tested compounds, OA-4 (3.13 µg/mL) showed significant activity against the secretion of HBsAg, HBeAg, and HBV DNA replication with inhibitory ratios of 90.52% ± 1.78%, 31.55% ± 3.65%, and 94.57% ± 3.11% after 6 days, respectively. Besides, OA-4 was further investigated in a duck model with DHBV infection. When OA-4 was administered at a dosage of 500 mg/kg, the results revealed a significant inhibitory effects of DHBV at 19.94% ± 2.87%, 28.80% ± 3.62% and 29.25% ± 2.65% at days 5, 10, and 3 after the cessation of OA-4 treatment, respectively. It's worth noting that OA-4 is superior to lamivudine in the inhibition of rebound of viral replication rate. The structure-activity relationships of OA derivatives had been preliminary discussed, which should be useful to explore further novel anti-HBV agents.
Keywords: anti-hepatitis B virus; eosin Y; epoxy ring; oleanolic-acid derivatives; virus recrudescence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Principal Bioactive Properties of Oleanolic Acid, Its Derivatives, and Analogues.Molecules. 2024 Jul 12;29(14):3291. doi: 10.3390/molecules29143291. Molecules. 2024. PMID: 39064870 Free PMC article. Review.
-
Anti-hepatitis B virus activity of wogonin in vitro and in vivo.Antiviral Res. 2007 Apr;74(1):16-24. doi: 10.1016/j.antiviral.2007.01.002. Epub 2007 Jan 24. Antiviral Res. 2007. PMID: 17280723
-
In vitro and in vivo anti-hepatitis B virus activities of the lignan nirtetralin B isolated from Phyllanthus niruri L.J Ethnopharmacol. 2014 Nov 18;157:62-8. doi: 10.1016/j.jep.2014.09.019. Epub 2014 Sep 27. J Ethnopharmacol. 2014. PMID: 25260580
-
Effect of various pyrimidines possessing the 1-[(2-hydroxy-1-(hydroxymethyl)ethoxy)methyl] moiety, able to mimic natural 2'-deoxyribose, on wild-type and mutant hepatitis B virus replication.J Med Chem. 2006 Jun 15;49(12):3693-700. doi: 10.1021/jm060102l. J Med Chem. 2006. PMID: 16759112
-
Small-molecule inhibitors for the treatment of hepatitis B virus documented in patents.Mini Rev Med Chem. 2013 Apr 1;13(5):749-76. doi: 10.2174/1389557511313050012. Mini Rev Med Chem. 2013. PMID: 23317498 Review.
Cited by
-
Principal Bioactive Properties of Oleanolic Acid, Its Derivatives, and Analogues.Molecules. 2024 Jul 12;29(14):3291. doi: 10.3390/molecules29143291. Molecules. 2024. PMID: 39064870 Free PMC article. Review.
-
Therapeutic potential of oleanolic acid in liver diseases.Naunyn Schmiedebergs Arch Pharmacol. 2024 Jul;397(7):4537-4554. doi: 10.1007/s00210-024-02959-2. Epub 2024 Jan 31. Naunyn Schmiedebergs Arch Pharmacol. 2024. PMID: 38294504 Review.
-
Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview.Pharmaceuticals (Basel). 2021 Apr 19;14(4):381. doi: 10.3390/ph14040381. Pharmaceuticals (Basel). 2021. PMID: 33921724 Free PMC article. Review.
-
Construction, structural modification, and bioactivity evaluation of pentacyclic triterpenoid privileged scaffolds in active natural products.RSC Adv. 2024 Dec 13;14(53):39436-39461. doi: 10.1039/d4ra07602h. eCollection 2024 Dec 10. RSC Adv. 2024. PMID: 39679419 Free PMC article. Review.
-
Antiviral Activities of Oleanolic Acid and Its Analogues.Molecules. 2018 Sep 9;23(9):2300. doi: 10.3390/molecules23092300. Molecules. 2018. PMID: 30205592 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

