β-Lactam Antibiotics Renaissance
- PMID: 27025744
- PMCID: PMC4790388
- DOI: 10.3390/antibiotics3020193
β-Lactam Antibiotics Renaissance
Abstract
Since the 1940s β-lactam antibiotics have been used to treat bacterial infections. However, emergence and dissemination of β-lactam resistance has reached the point where many marketed β-lactams no longer are clinically effective. The increasing prevalence of multidrug-resistant bacteria and the progressive withdrawal of pharmaceutical companies from antibiotic research have evoked a strong reaction from health authorities, who have implemented initiatives to encourage the discovery of new antibacterials. Despite this gloomy scenario, several novel β-lactam antibiotics and β-lactamase inhibitors have recently progressed into clinical trials, and many more such compounds are being investigated. Here we seek to provide highlights of recent developments relating to the discovery of novel β-lactam antibiotics and β-lactamase inhibitors.
Keywords: bacterial infections; β-lactam antibiotics; β-lactamase inhibitors.
Figures
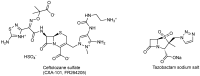
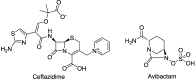

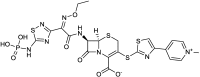
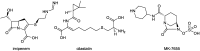
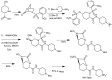
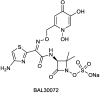

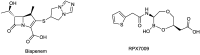

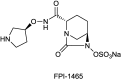
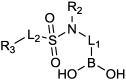
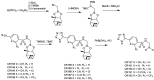
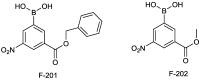


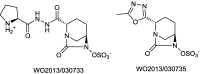
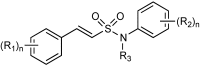
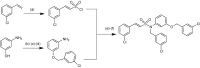

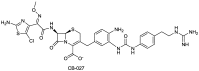
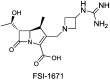
References
-
- Shlaes D.M. Antibiotics: The Perfect Storm. Springer Dordrec; Heilderberg, London, NY, USA: 2010.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

