Lipopolysaccharide transport and assembly at the outer membrane: the PEZ model
- PMID: 27026255
- PMCID: PMC4937791
- DOI: 10.1038/nrmicro.2016.25
Lipopolysaccharide transport and assembly at the outer membrane: the PEZ model
Abstract
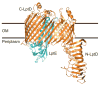
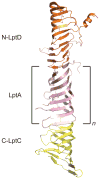
Gram-negative bacteria have a double-membrane cellular envelope that enables them to colonize harsh environments and prevents the entry of many clinically available antibiotics. A main component of most outer membranes is lipopolysaccharide (LPS), a glycolipid containing several fatty acyl chains and up to hundreds of sugars that is synthesized in the cytoplasm. In the past two decades, the proteins that are responsible for transporting LPS across the cellular envelope and assembling it at the cell surface in Escherichia coli have been identified, but it remains unclear how they function. In this Review, we discuss recent advances in this area and present a model that explains how energy from the cytoplasm is used to power LPS transport across the cellular envelope to the cell surface.
Conflict of interest statement
I declare that the authors have no competing interests as defined by Nature Publishing Group, or other interests that might be perceived to influence the results and discussion reported in this paper.
Figures



References
-
- Berg HC. The rotary motor of bacterial flagella. Annu Rev Biochem. 2003;72:19–54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

