Metabotropic Glutamate Receptors and Interacting Proteins in Epileptogenesis
- PMID: 27030135
- PMCID: PMC4983745
- DOI: 10.2174/1570159x14666160331142228
Metabotropic Glutamate Receptors and Interacting Proteins in Epileptogenesis
Abstract
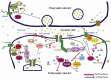
Neurotransmitter and receptor systems are involved in different neurological and neuropsychological disorders such as Parkinson's disease, depression, Alzheimer's disease and epilepsy. Recent advances in studies of signal transduction pathways or interacting proteins of neurotransmitter receptor systems suggest that different receptor systems may share the common signal transduction pathways or interacting proteins which may be better therapeutic targets for development of drugs to effectively control brain diseases. In this paper, we reviewed metabotropic glutamate receptors (mGluRs) and their related signal transduction pathways or interacting proteins in status epilepticus and temporal lobe epilepsy, and proposed some novel therapeutical drug targets for controlling epilepsy and epileptogenesis.
Figures
Similar articles
-
Metabotropic glutamate receptors as drug targets.Curr Drug Targets. 2007 May;8(5):651-81. doi: 10.2174/138945007780618544. Curr Drug Targets. 2007. PMID: 17504108 Review.
-
Metabotropic glutamate receptors and interacting proteins: evolving drug targets.Curr Drug Targets. 2012 Jan;13(1):145-56. doi: 10.2174/138945012798868452. Curr Drug Targets. 2012. PMID: 21777188 Review.
-
Modulation of calcium channels by group I and group II metabotropic glutamate receptors in dentate gyrus neurons from patients with temporal lobe epilepsy.Epilepsia. 2000 Oct;41(10):1249-58. doi: 10.1111/j.1528-1157.2000.tb04602.x. Epilepsia. 2000. PMID: 11051119
-
Metabotropic glutamate receptors in the control of neuronal activity and as targets for development of anti-epileptogenic drugs.Curr Med Chem. 2009;16(17):2189-204. doi: 10.2174/092986709788612710. Curr Med Chem. 2009. PMID: 19519386 Review.
-
Glutamate receptor involvement in dentate granule cell epileptiform activity evoked by mossy fiber stimulation.Brain Res. 2001 Oct 5;915(1):58-69. doi: 10.1016/s0006-8993(01)02824-4. Brain Res. 2001. PMID: 11578620
Cited by
-
microRNA-182 Negatively Influences the Neuroprotective Effect of Apelin Against Neuronal Injury in Epilepsy.Neuropsychiatr Dis Treat. 2020 Jan 30;16:327-338. doi: 10.2147/NDT.S238826. eCollection 2020. Neuropsychiatr Dis Treat. 2020. PMID: 32099369 Free PMC article.
-
Novel Targets for Developing Antiseizure and, Potentially, Antiepileptogenic Drugs.Epilepsy Curr. 2017 Sep-Oct;17(5):293-298. doi: 10.5698/1535-7597.17.5.293. Epilepsy Curr. 2017. PMID: 29225544 Free PMC article.
-
Substance P Regulation in Epilepsy.Curr Neuropharmacol. 2018;16(1):43-50. doi: 10.2174/1570159X15666170504122410. Curr Neuropharmacol. 2018. PMID: 28474564 Free PMC article. Review.
-
The Influence of Vaccine on Febrile Seizure.Curr Neuropharmacol. 2018;16(1):59-65. doi: 10.2174/1570159X15666170726115639. Curr Neuropharmacol. 2018. PMID: 28745219 Free PMC article. Review.
-
Dysregulation of Ambient Glutamate and Glutamate Receptors in Epilepsy: An Astrocytic Perspective.Front Neurol. 2021 Mar 22;12:652159. doi: 10.3389/fneur.2021.652159. eCollection 2021. Front Neurol. 2021. PMID: 33828523 Free PMC article. Review.
References
-
- Enz R. Metabotropic glutamate receptors and interacting proteins: evolving drug targets. Curr. Drug Targets. 2012;13(1):145–156. [http://dx.doi.org/10.2174/138945012798868452]. [PMID: 21777188]. - PubMed
-
- Sladeczek F., Pin J.P., Récasens M., Bockaert J., Weiss S. Glutamate stimulates inositol phosphate formation in striatal neurones. Nature. 1985;317(6039):717–719. [http://dx.doi.org/ 10.1038/317717a0]. [PMID: 2865680]. - PubMed
-
- Cotman C.W., Flatman J.A., Ganong A.H., Perkins M.N. Effects of excitatory amino acid antagonists on evoked and spontaneous excitatory potentials in guinea-pig hippocampus. J. Physiol. 1986;378:403–415. [http://dx.doi.org/10.1113/jphysiol.1986.sp016227]. [PMID: 3795109]. - PMC - PubMed
-
- Masu M., Tanabe Y., Tsuchida K., Shigemoto R., Nakanishi S. Sequence and expression of a metabotropic glutamate receptor. Nature. 1991;349(6312):760–765. [http://dx.doi.org/10.1038/ 349760a0]. [PMID: 1847995]. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
