Targeting of tubulin polymerization and induction of mitotic blockage by Methyl 2-(5-fluoro-2-hydroxyphenyl)-1H-benzo[d]imidazole-5-carboxylate (MBIC) in human cervical cancer HeLa cell
- PMID: 27030360
- PMCID: PMC4815073
- DOI: 10.1186/s13046-016-0332-0
Targeting of tubulin polymerization and induction of mitotic blockage by Methyl 2-(5-fluoro-2-hydroxyphenyl)-1H-benzo[d]imidazole-5-carboxylate (MBIC) in human cervical cancer HeLa cell
Abstract
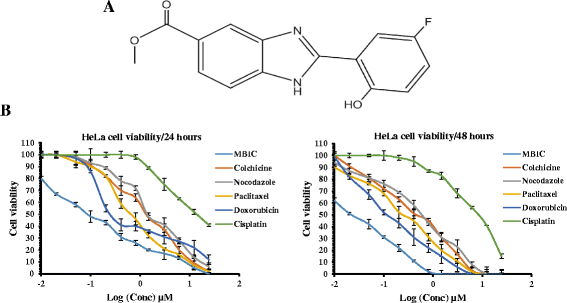
Background: Microtubule Targeting Agents (MTAs) including paclitaxel, colchicine and vinca alkaloids are widely used in the treatment of various cancers. As with most chemotherapeutic agents, adverse effects and drug resistance are commonly associated with the clinical use of these agents. Methyl 2-(5-fluoro-2-hydroxyphenyl)-1H- benzo[d]imidazole-5-carboxylate (MBIC), a benzimidazole derivative displays greater toxicity against various cancer compared to normal human cell lines. The present study, focused on the cytotoxic effects of MBIC against HeLa cervical cancer cells and possible actions on the microtubule assembly.
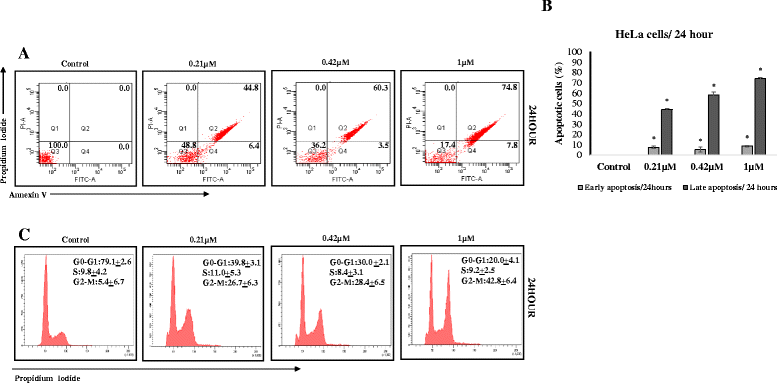
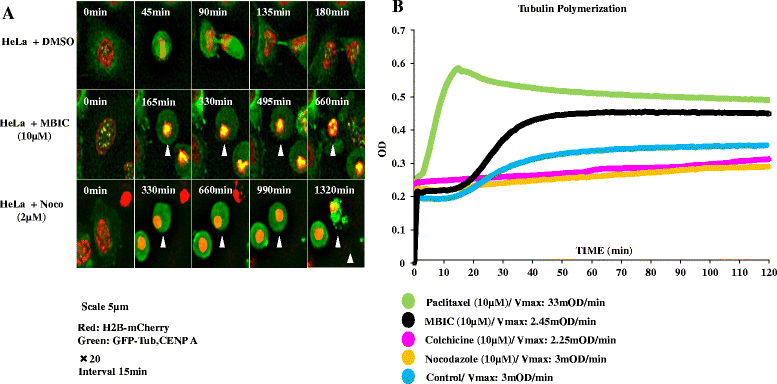
Methods: Apoptosis detection and cell-cycle assays were performed to determine the type of cell death and the phase of cell cycle arrest in HeLa cells. Tubulin polymerization assay and live-cell imaging were performed to visualize effects on the microtubule assembly in the presence of MBIC. Mitotic kinases and mitochondrial-dependent apoptotic proteins were evaluated by Western blot analysis. In addition, the synergistic effect of MBIC with low doses of selected chemotherapeutic actions were examined against the cancer cells.
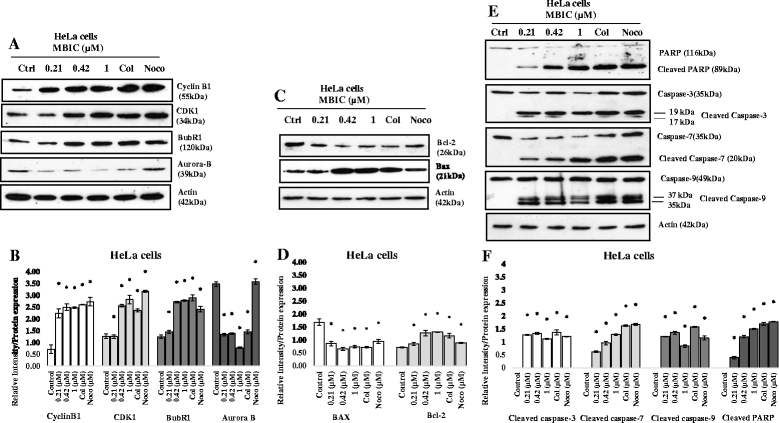
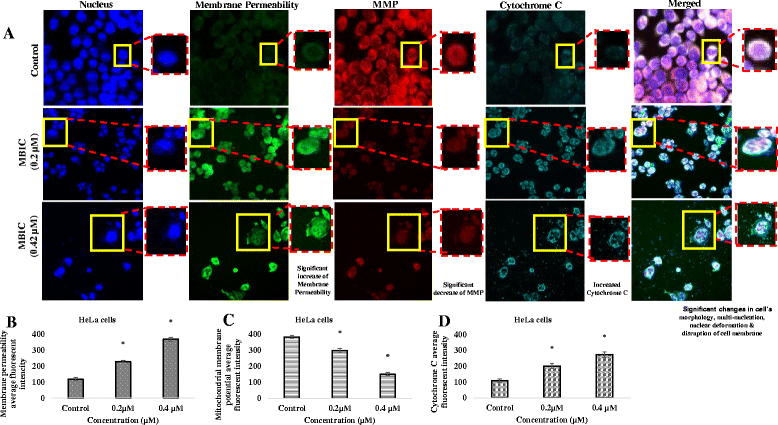
Results: Results from the present study showed that following treatment with MBIC, the HeLa cells went into mitotic arrest comprising of multi-nucleation and unsegregated chromosomes with a prolonged G2-M phase. In addition, the HeLa cells showed signs of mitochondrial-dependant apoptotic features such as the release of cytochrome c and activation of caspases. MBIC markedly interferes with tubulin polymerization. Western blotting results indicated that MBIC affects mitotic regulatory machinery by up-regulating BubR1, Cyclin B1, CDK1 and down-regulation of Aurora B. In addition, MBIC displayed synergistic effect when given in combination with colchicine, nocodazole, paclitaxel and doxorubicin.
Conclusion: Taken together, our study demonstrated the distinctive microtubule destabilizing effects of MBIC against cervical cancer cells in vitro. Besides that, MBIC exhibited synergistic effects with low doses of selected anticancer drugs and thus, may potentially reduce the toxicity and drug resistance to these agents.
Keywords: Cervical cancer; Microtubule dynamics; Mitochondrial-dependent apoptosis; Mitotic arrest.
Figures

Similar articles
-
Mechanisms of the anti-tumor activity of Methyl 2-(-5-fluoro-2-hydroxyphenyl)-1 H-benzo[d]imidazole-5-carboxylate against breast cancer in vitro and in vivo.Oncotarget. 2017 Apr 25;8(17):28840-28853. doi: 10.18632/oncotarget.16263. Oncotarget. 2017. PMID: 28392503 Free PMC article.
-
6,7-Dimethoxy-3-(3-methoxyphenyl)isoquinolin-1-amine induces mitotic arrest and apoptotic cell death through the activation of spindle assembly checkpoint in human cervical cancer cells.Carcinogenesis. 2013 Aug;34(8):1852-60. doi: 10.1093/carcin/bgt133. Epub 2013 Apr 24. Carcinogenesis. 2013. PMID: 23615402
-
Methyl 5-[(1H-indol-3-yl)selanyl]-1H-benzoimidazol-2-ylcarbamate (M-24), a novel tubulin inhibitor, causes G2/M arrest and cell apoptosis by disrupting tubulin polymerization in human cervical and breast cancer cells.Toxicol In Vitro. 2017 Aug;42:139-149. doi: 10.1016/j.tiv.2017.04.019. Epub 2017 Apr 13. Toxicol In Vitro. 2017. PMID: 28412508
-
Microtubules and Cell Division: Potential Pharmacological Targets in Cancer Therapy.Curr Drug Targets. 2023;24(11):889-918. doi: 10.2174/1389450124666230731094837. Curr Drug Targets. 2023. PMID: 37519203 Review.
-
[Inhibitors of microtubule polymerization- new natural compounds as potential anti-cancer drugs].Postepy Hig Med Dosw (Online). 2015 May 4;69:571-85. doi: 10.5604/17322693.1151293. Postepy Hig Med Dosw (Online). 2015. PMID: 25983296 Review. Polish.
Cited by
-
Mechanisms of the anti-tumor activity of Methyl 2-(-5-fluoro-2-hydroxyphenyl)-1 H-benzo[d]imidazole-5-carboxylate against breast cancer in vitro and in vivo.Oncotarget. 2017 Apr 25;8(17):28840-28853. doi: 10.18632/oncotarget.16263. Oncotarget. 2017. PMID: 28392503 Free PMC article.
-
Design, synthesis, and in vitro antitumor evaluation of novel benzimidazole acylhydrazone derivatives.Mol Divers. 2025 Jan 18. doi: 10.1007/s11030-024-11064-8. Online ahead of print. Mol Divers. 2025. PMID: 39825985
-
Benzimidazole and its derivatives as cancer therapeutics: The potential role from traditional to precision medicine.Acta Pharm Sin B. 2023 Feb;13(2):478-497. doi: 10.1016/j.apsb.2022.09.010. Epub 2022 Sep 21. Acta Pharm Sin B. 2023. PMID: 36873180 Free PMC article. Review.
-
Treatment with a new benzimidazole derivative bearing a pyrrolidine side chain overcomes sorafenib resistance in hepatocellular carcinoma.Sci Rep. 2019 Nov 21;9(1):17259. doi: 10.1038/s41598-019-53863-2. Sci Rep. 2019. PMID: 31754201 Free PMC article.
-
Anticancer Effect of PtIIPHENSS, PtII5MESS, PtII56MESS and Their Platinum(IV)-Dihydroxy Derivatives against Triple-Negative Breast Cancer and Cisplatin-Resistant Colorectal Cancer.Cancers (Basel). 2024 Jul 15;16(14):2544. doi: 10.3390/cancers16142544. Cancers (Basel). 2024. PMID: 39061185 Free PMC article.
References
-
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA: a cancer journal for clinicians. 2011;61(2):69–90. - PubMed
-
- Cadron I, Amant F, Vergote I. Chemotherapy for cervical cancer. European Clinics in Obstetrics and Gynaecology. 2005;1(3):143–150. doi: 10.1007/s11296-005-0006-5. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

