Major spliceosome defects cause male infertility and are associated with nonobstructive azoospermia in humans
- PMID: 27035939
- PMCID: PMC4839444
- DOI: 10.1073/pnas.1513682113
Major spliceosome defects cause male infertility and are associated with nonobstructive azoospermia in humans
Abstract
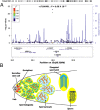
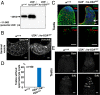
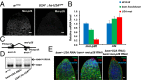
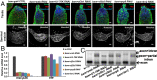
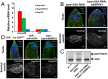
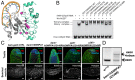
Processing of pre-mRNA into mRNA is an important regulatory mechanism in eukaryotes that is mediated by the spliceosome, a huge and dynamic ribonucleoprotein complex. Splicing defects are implicated in a spectrum of human disease, but the underlying mechanistic links remain largely unresolved. Using a genome-wide association approach, we have recently identified single nucleotide polymorphisms in humans that associate with nonobstructive azoospermia (NOA), a common cause of male infertility. Here, using genetic manipulation of corresponding candidate loci in Drosophila, we show that the spliceosome component SNRPA1/U2A is essential for male fertility. Loss of U2A in germ cells of the Drosophila testis does not affect germline stem cells, but does result in the accumulation of mitotic spermatogonia that fail to differentiate into spermatocytes and mature sperm. Lack of U2A causes insufficient splicing of mRNAs required for the transition of germ cells from proliferation to differentiation. We show that germ cell-specific disruption of other components of the major spliceosome manifests with the same phenotype, demonstrating that mRNA processing is required for the differentiation of spermatogonia. This requirement is conserved, and expression of human SNRPA1 fully restores spermatogenesis in U2A mutant flies. We further report that several missense mutations in human SNRPA1 that inhibit the assembly of the major spliceosome dominantly disrupt spermatogonial differentiation in Drosophila. Collectively, our findings uncover a conserved and specific requirement for the major spliceosome during the transition from spermatogonial proliferation to differentiation in the male testis, suggesting that spliceosome defects affecting the differentiation of human spermatogonia contribute to NOA.
Keywords: GWAS; NOA; spermatogenesis; spermatogonia; spliceosome.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Precursor RNA processing 3 is required for male fertility, and germline stem cell self-renewal and differentiation via regulating spliceosome function in Drosophila testes.Sci Rep. 2019 Jul 10;9(1):9988. doi: 10.1038/s41598-019-46419-x. Sci Rep. 2019. PMID: 31292463 Free PMC article.
-
Restore natural fertility of Kitw/Kitwv mouse with nonobstructive azoospermia through gene editing on SSCs mediated by CRISPR-Cas9.Stem Cell Res Ther. 2019 Aug 24;10(1):271. doi: 10.1186/s13287-019-1386-7. Stem Cell Res Ther. 2019. PMID: 31445521 Free PMC article.
-
Investigation of CD133 and CD24 as candidate azoospermia markers and their relationship with spermatogenesis defects.Gene. 2019 Jul 20;706:211-221. doi: 10.1016/j.gene.2019.04.028. Epub 2019 May 1. Gene. 2019. PMID: 31054360
-
Progress in the genetic studies of spermatogenesis abnormalities.Yi Chuan. 2021 May 20;43(5):473-486. doi: 10.16288/j.yczz.20-343. Yi Chuan. 2021. PMID: 33972217 Review.
-
In vitro spermatogenesis in artificial testis: current knowledge and clinical implications for male infertility.Cell Tissue Res. 2023 Dec;394(3):393-421. doi: 10.1007/s00441-023-03824-z. Epub 2023 Sep 18. Cell Tissue Res. 2023. PMID: 37721632 Review.
Cited by
-
Splicing regulation in brain and testis: common themes for highly specialized organs.Cell Cycle. 2021 Mar-Mar;20(5-6):480-489. doi: 10.1080/15384101.2021.1889187. Epub 2021 Feb 26. Cell Cycle. 2021. PMID: 33632061 Free PMC article. Review.
-
Effect of Dimethylacetamide Concentration on Motility, Quality, Antioxidant Biomarkers, Anti-Freeze Gene Expression, and Fertilizing Ability of Frozen/Thawed Rooster Sperm.Animals (Basel). 2022 Oct 12;12(20):2739. doi: 10.3390/ani12202739. Animals (Basel). 2022. PMID: 36290126 Free PMC article.
-
Comparative transcriptome analysis digs out genes related to antifreeze between fresh and frozen-thawed rooster sperm.Poult Sci. 2020 Jun;99(6):2841-2851. doi: 10.1016/j.psj.2020.01.022. Epub 2020 Mar 19. Poult Sci. 2020. PMID: 32475417 Free PMC article.
-
The In Vitro Evaluation of Rooster Semen Pellets Frozen with Dimethylacetamide.Animals (Basel). 2023 May 11;13(10):1603. doi: 10.3390/ani13101603. Animals (Basel). 2023. PMID: 37238034 Free PMC article.
-
The Role of Genetics and Oxidative Stress in the Etiology of Male Infertility-A Unifying Hypothesis?Front Endocrinol (Lausanne). 2020 Sep 30;11:581838. doi: 10.3389/fendo.2020.581838. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 33101214 Free PMC article. Review.
References
-
- Lee JY, Dada R, Sabanegh E, Carpi A, Agarwal A. Role of genetics in azoospermia. Urology. 2011;77(3):598–601. - PubMed
-
- Ferlin A, Arredi B, Foresta C. Genetic causes of male infertility. Reprod Toxicol. 2006;22(2):133–141. - PubMed
-
- Dieterich K, et al. Homozygous mutation of AURKC yields large-headed polyploid spermatozoa and causes male infertility. Nat Genet. 2007;39(5):661–665. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

