Dopamine synapse is a neuroligin-2-mediated contact between dopaminergic presynaptic and GABAergic postsynaptic structures
- PMID: 27035941
- PMCID: PMC4839454
- DOI: 10.1073/pnas.1514074113
Dopamine synapse is a neuroligin-2-mediated contact between dopaminergic presynaptic and GABAergic postsynaptic structures
Abstract
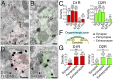
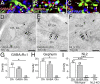
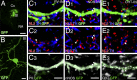
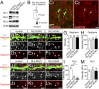
Midbrain dopamine neurons project densely to the striatum and form so-called dopamine synapses on medium spiny neurons (MSNs), principal neurons in the striatum. Because dopamine receptors are widely expressed away from dopamine synapses, it remains unclear how dopamine synapses are involved in dopaminergic transmission. Here we demonstrate that dopamine synapses are contacts formed between dopaminergic presynaptic and GABAergic postsynaptic structures. The presynaptic structure expressed tyrosine hydroxylase, vesicular monoamine transporter-2, and plasmalemmal dopamine transporter, which are essential for dopamine synthesis, vesicular filling, and recycling, but was below the detection threshold for molecules involving GABA synthesis and vesicular filling or for GABA itself. In contrast, the postsynaptic structure of dopamine synapses expressed GABAergic molecules, including postsynaptic adhesion molecule neuroligin-2, postsynaptic scaffolding molecule gephyrin, and GABAA receptor α1, without any specific clustering of dopamine receptors. Of these, neuroligin-2 promoted presynaptic differentiation in axons of midbrain dopamine neurons and striatal GABAergic neurons in culture. After neuroligin-2 knockdown in the striatum, a significant decrease of dopamine synapses coupled with a reciprocal increase of GABAergic synapses was observed on MSN dendrites. This finding suggests that neuroligin-2 controls striatal synapse formation by giving competitive advantage to heterologous dopamine synapses over conventional GABAergic synapses. Considering that MSN dendrites are preferential targets of dopamine synapses and express high levels of dopamine receptors, dopamine synapse formation may serve to increase the specificity and potency of dopaminergic modulation of striatal outputs by anchoring dopamine release sites to dopamine-sensing targets.
Keywords: dopamine synapse; medium spiny neuron; neuroligin-2; striatum.
Conflict of interest statement
The authors declare no conflict of interest.
Figures









Similar articles
-
Differential role of GABAA receptors and neuroligin 2 for perisomatic GABAergic synapse formation in the hippocampus.Brain Struct Funct. 2017 Dec;222(9):4149-4161. doi: 10.1007/s00429-017-1462-7. Epub 2017 Jun 22. Brain Struct Funct. 2017. PMID: 28643105
-
Distinct mechanisms regulate GABAA receptor and gephyrin clustering at perisomatic and axo-axonic synapses on CA1 pyramidal cells.J Physiol. 2011 Oct 15;589(Pt 20):4959-80. doi: 10.1113/jphysiol.2011.216028. Epub 2011 Aug 8. J Physiol. 2011. PMID: 21825022 Free PMC article.
-
Dopaminergic treatment weakens medium spiny neuron collateral inhibition in the parkinsonian striatum.J Neurophysiol. 2017 Mar 1;117(3):987-999. doi: 10.1152/jn.00683.2016. Epub 2016 Dec 7. J Neurophysiol. 2017. PMID: 27927785 Free PMC article.
-
GABA and neuroligin signaling: linking synaptic activity and adhesion in inhibitory synapse development.Curr Opin Neurobiol. 2008 Feb;18(1):77-83. doi: 10.1016/j.conb.2008.05.008. Epub 2008 May 29. Curr Opin Neurobiol. 2008. PMID: 18513949 Free PMC article. Review.
-
Neuroligins, synapse balance and neuropsychiatric disorders.Pharmacol Rep. 2014 Oct;66(5):830-5. doi: 10.1016/j.pharep.2014.04.011. Epub 2014 May 9. Pharmacol Rep. 2014. PMID: 25149987 Review.
Cited by
-
The differentially expressed proteins related to clinical viral encephalitis revealed by proteomics.Ibrain. 2022 May 4;8(2):148-164. doi: 10.1002/ibra.12036. eCollection 2022 Summer. Ibrain. 2022. PMID: 37786892 Free PMC article.
-
A novel synaptic junction preparation for the identification and characterization of cleft proteins.PLoS One. 2017 Mar 31;12(3):e0174895. doi: 10.1371/journal.pone.0174895. eCollection 2017. PLoS One. 2017. PMID: 28362857 Free PMC article.
-
Loss of Neuroligin3 specifically downregulates retinal GABAAα2 receptors without abolishing direction selectivity.PLoS One. 2017 Jul 14;12(7):e0181011. doi: 10.1371/journal.pone.0181011. eCollection 2017. PLoS One. 2017. PMID: 28708891 Free PMC article.
-
Distinct mechanisms control the specific synaptic functions of Neuroligin 1 and Neuroligin 2.EMBO Rep. 2025 Feb;26(3):860-879. doi: 10.1038/s44319-024-00286-4. Epub 2025 Jan 2. EMBO Rep. 2025. PMID: 39747663 Free PMC article.
-
GABA uptake transporters support dopamine release in dorsal striatum with maladaptive downregulation in a parkinsonism model.Nat Commun. 2020 Oct 2;11(1):4958. doi: 10.1038/s41467-020-18247-5. Nat Commun. 2020. PMID: 33009395 Free PMC article.
References
-
- Lardi-Studler B, Fritschy JM. Matching of pre- and postsynaptic specializations during synaptogenesis. Neuroscientist. 2007;13(2):115–126. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

