Rethinking progesterone regulation of female reproductive cyclicity
- PMID: 27035990
- PMCID: PMC4839436
- DOI: 10.1073/pnas.1601825113
Rethinking progesterone regulation of female reproductive cyclicity
Abstract
The progesterone receptor (PGR) is a ligand-activated transcription factor with key roles in the regulation of female fertility. Much has been learned of the actions of PGR signaling through the use of pharmacologic inhibitors and genetic manipulation, using mouse mutagenesis. Characterization of rats with a null mutation at the Pgr locus has forced a reexamination of the role of progesterone in the regulation of the female reproductive cycle. We generated two Pgr mutant rat models, using genome editing. In both cases, deletions yielded a null mutation resulting from a nonsense frame-shift and the emergence of a stop codon. Similar to Pgr null mice, Pgr null rats were infertile because of deficits in sexual behavior, ovulation, and uterine endometrial differentiation. However, in contrast to the reported phenotype of female mice with disruptions in Pgr signaling, Pgr null female rats exhibit robust estrous cycles. Cyclic changes in vaginal cytology, uterine histology, serum hormone levels, and wheel running activity were evident in Pgr null female rats, similar to wild-type controls. Furthermore, exogenous progesterone treatment inhibited estrous cycles in wild-type female rats but not in Pgr-null female rats. As previously reported, pharmacologic antagonism supports a role for PGR signaling in the regulation of the ovulatory gonadotropin surge, a result at variance with experimentation using genetic ablation of PGR signaling. To conclude, our findings in the Pgr null rat challenge current assumptions and prompt a reevaluation of the hormonal control of reproductive cyclicity.
Keywords: PGR; progesterone; rat; reproductive cycles.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


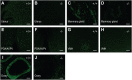



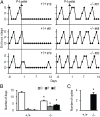



References
-
- Beshay VE, Carr BR. Hypothalamic-pituitary-ovarian axis and control of the menstrual cycle. In: Falcone T, Hurd WW, editors. Clinical Reproductive Medicine and Surgery: A Practical Guide. Springer; New York: 2012. pp. 31–42.
-
- Goodman RL, Karsch FJ. Pulsatile secretion of luteinizing hormone: Differential suppression by ovarian steroids. Endocrinology. 1980;107(5):1286–1290. - PubMed
-
- Plant TM. Gonadal regulation of hypothalamic gonadotropin-releasing hormone release in primates. Endocr Rev. 1986;7(1):75–88. - PubMed
-
- Clarke IJ. Control of GnRH secretion: One step back. Front Neuroendocrinol. 2011;32(3):367–375. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

