NanoLuc: A Small Luciferase Is Brightening Up the Field of Bioluminescence
- PMID: 27045664
- PMCID: PMC4871753
- DOI: 10.1021/acs.bioconjchem.6b00112
NanoLuc: A Small Luciferase Is Brightening Up the Field of Bioluminescence
Abstract
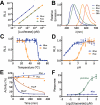
The biomedical field has greatly benefited from the discovery of bioluminescent proteins. Currently, scientists employ bioluminescent systems for numerous biomedical applications, ranging from highly sensitive cellular assays to bioluminescence-based molecular imaging. Traditionally, these systems are based on Firefly and Renilla luciferases; however, the applicability of these enzymes is limited by their size, stability, and luminescence efficiency. NanoLuc (NLuc), a novel bioluminescence platform, offers several advantages over established systems, including enhanced stability, smaller size, and >150-fold increase in luminescence. In addition, the substrate for NLuc displays enhanced stability and lower background activity, opening up new possibilities in the field of bioluminescence imaging. The NLuc system is incredibly versatile and may be utilized for a wide array of applications. The increased sensitivity, high stability, and small size of the NLuc system have the potential to drastically change the field of reporter assays in the future. However, as with all such technology, NLuc has limitations (including a nonideal emission for in vivo applications and its unique substrate) which may cause it to find restricted use in certain areas of molecular biology. As this unique technology continues to broaden, NLuc may have a significant impact in both preclinical and clinical fields, with potential roles in disease detection, molecular imaging, and therapeutic monitoring. This review will present the NLuc technology to the scientific community in a nonbiased manner, allowing the audience to adopt their own views of this novel system.
Figures






References
-
- Widder EA, Falls B. Review of Bioluminescence for Engineers and Scientists in Biophotonics. IEEE J. Sel. Top. Quantum Electron. 2014;20
-
- Roda A. Chemiluminescence and Bioluminescence: Past, Present and Future. The Royal Society of Chemistry; 2011. hapter 1 A History of Bioluminescence and Chemiluminescence from Ancient Times to the Present; pp. 1–50.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

