Optogenetic approaches addressing extracellular modulation of neural excitability
- PMID: 27045897
- PMCID: PMC4820717
- DOI: 10.1038/srep23947
Optogenetic approaches addressing extracellular modulation of neural excitability
Abstract
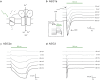
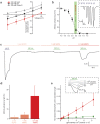
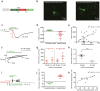
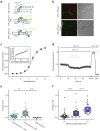
The extracellular ionic environment in neural tissue has the capacity to influence, and be influenced by, natural bouts of neural activity. We employed optogenetic approaches to control and investigate these interactions within and between cells, and across spatial scales. We began by developing a temporally precise means to study microdomain-scale interactions between extracellular protons and acid-sensing ion channels (ASICs). By coupling single-component proton-transporting optogenetic tools to ASICs to create two-component optogenetic constructs (TCOs), we found that acidification of the local extracellular membrane surface by a light-activated proton pump recruited a slow inward ASIC current, which required molecular proximity of the two components on the membrane. To elicit more global effects of activity modulation on 'bystander' neurons not under direct control, we used densely-expressed depolarizing (ChR2) or hyperpolarizing (eArch3.0, eNpHR3.0) tools to create a slow non-synaptic membrane current in bystander neurons, which matched the current direction seen in the directly modulated neurons. Extracellular protons played contributory role but were insufficient to explain the entire bystander effect, suggesting the recruitment of other mechanisms. Together, these findings present a new approach to the engineering of multicomponent optogenetic tools to manipulate ionic microdomains, and probe the complex neuronal-extracellular space interactions that regulate neural excitability.
Figures

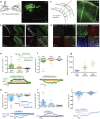
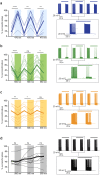

References
-
- Poolos N. P., Mauk M. D. & Kocsis J. D. Activity-Evoked Increases in Extracellular Potassium Modulate Presynaptic Excitability in the CA1 Region of the Hippocampus. J. Neurophysiol. 58, 404–416 (1987). - PubMed
-
- Krishtal O. A., Osipchuk Y. V., Shelest T. N. & Smirnoff S. V. Rapid extracellular pH transients related to synaptic transmission in rat hippocampal slices. Brain Res. 436, 352–356 (1987). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

