The role of bile salts in liver regeneration
- PMID: 27048617
- PMCID: PMC5003899
- DOI: 10.1007/s12072-016-9723-8
The role of bile salts in liver regeneration
Abstract
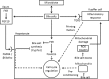
A growing body of evidence has demonstrated that bile salts are important for liver regeneration following partial hepatectomy. The relative bile salt overload after partial liver resection causes activation of bile salt receptors in non-parenchymal (viz. the plasma membrane receptor TGR5) and parenchymal (viz. the intracellular receptor FXR) cells in the liver, thus, providing signals to the regenerative process. Impaired bile salt signaling in mice with genetic deficiency of Tgr5 or Fxr results in delayed liver regeneration after partial hepatectomy, and is accompanied by mortality in case of Fxr knock-out mice. Conversely, compensatory liver re-growth in hepatectomized mice can be stimulated by feeding of bile salts or alisol B 23-acetate, a natural triterpenoid agonist of Fxr. A large number of animal studies underscore the importance of strict maintenance of bile salt homeostasis for proper progression of liver regeneration. Both ileal and hepatic Fxr play a key role in regulation of bile salt homeostasis and, thus, preventing hepatotoxicity caused by excessive levels of bile salts. They further contribute to liver regeneration by induction of mitogenic factors. Agents that target bile salt receptors hold promise as drugs to stimulate liver regeneration in selected patients.
Keywords: Bile salt homeostasis; Bile salt signaling; Farnesoid X receptor; Liver regeneration; Liver surgery.
Conflict of interest statement
Compliance with ethical standards All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors. Conflict of interest Liyanne van de Laarschot, Peter Jansen, Frank Schaap, and Steven Olde Damink declare that they have no conflict of interest.
Figures
Similar articles
-
Bile acids and their receptors during liver regeneration: "Dangerous protectors".Mol Aspects Med. 2017 Aug;56:25-33. doi: 10.1016/j.mam.2017.03.002. Epub 2017 Mar 23. Mol Aspects Med. 2017. PMID: 28302491 Review.
-
The receptor TGR5 protects the liver from bile acid overload during liver regeneration in mice.Hepatology. 2013 Oct;58(4):1451-60. doi: 10.1002/hep.26463. Epub 2013 Aug 19. Hepatology. 2013. PMID: 23686672
-
Nuclear bile acid receptor FXR in the hepatic regeneration.Biochim Biophys Acta. 2011 Aug;1812(8):888-92. doi: 10.1016/j.bbadis.2010.12.006. Epub 2010 Dec 15. Biochim Biophys Acta. 2011. PMID: 21167938 Review.
-
A role of the bile salt receptor FXR in atherosclerosis.Arterioscler Thromb Vasc Biol. 2010 Aug;30(8):1519-28. doi: 10.1161/ATVBAHA.109.197897. Arterioscler Thromb Vasc Biol. 2010. PMID: 20631352 Review.
-
Alisol B 23-acetate promotes liver regeneration in mice after partial hepatectomy via activating farnesoid X receptor.Biochem Pharmacol. 2014 Nov 15;92(2):289-98. doi: 10.1016/j.bcp.2014.09.009. Epub 2014 Sep 30. Biochem Pharmacol. 2014. PMID: 25278094
Cited by
-
Cholangiocarcinoma.Cancer Treat Res. 2024;192:165-184. doi: 10.1007/978-3-031-61238-1_9. Cancer Treat Res. 2024. PMID: 39212921 Review.
-
An Italian survey on the use of T-tube in liver transplantation: old habits die hard!Updates Surg. 2021 Aug;73(4):1381-1389. doi: 10.1007/s13304-021-01019-1. Epub 2021 Apr 1. Updates Surg. 2021. PMID: 33792888 Free PMC article.
-
Liver regeneration observed across the different classes of vertebrates from an evolutionary perspective.Heliyon. 2021 Mar 10;7(3):e06449. doi: 10.1016/j.heliyon.2021.e06449. eCollection 2021 Mar. Heliyon. 2021. PMID: 33748499 Free PMC article. Review.
-
Aminodihydrophthalazinedione Sodium Transdermal Therapeutic System Specific Activity on an ExperimentalModel of Extensive Liver Resection.Life (Basel). 2023 Feb 27;13(3):658. doi: 10.3390/life13030658. Life (Basel). 2023. PMID: 36983814 Free PMC article.
-
Circulating proliferative factors versus portal inflow redistribution: mechanistic insights of ALPPS-derived rapid liver regeneration.Front Oncol. 2025 Jan 7;14:1429564. doi: 10.3389/fonc.2024.1429564. eCollection 2024. Front Oncol. 2025. PMID: 39839786 Free PMC article. Review.
References
-
- Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology 2006;43(S1):S45–S53. doi:10.1002/hep.20969 - PubMed
-
- Higgins G, Anderson G. Experimental pathology of the liver restoration of the liver of the white rat following partial surgical removal. Arch Pathol Chicago 1931;12:186–202
-
- Makino H, Togo S, Kubota T, et al. A good model of hepatic failure after excessive hepatectomy in mice. J Surg Res 2005;127(2):171–176. doi:10.1016/j.jss.2005.04.029 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

