Mice with an NaV1.4 sodium channel null allele have latent myasthenia, without susceptibility to periodic paralysis
- PMID: 27048647
- PMCID: PMC4892753
- DOI: 10.1093/brain/aww070
Mice with an NaV1.4 sodium channel null allele have latent myasthenia, without susceptibility to periodic paralysis
Abstract
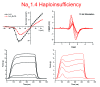
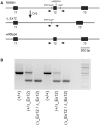
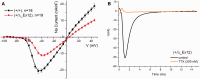
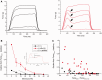
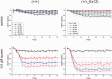
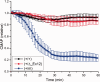
Over 60 mutations of SCN4A encoding the NaV1.4 sodium channel of skeletal muscle have been identified in patients with myotonia, periodic paralysis, myasthenia, or congenital myopathy. Most mutations are missense with gain-of-function defects that cause susceptibility to myotonia or periodic paralysis. Loss-of-function from enhanced inactivation or null alleles is rare and has been associated with myasthenia and congenital myopathy, while a mix of loss and gain of function changes has an uncertain relation to hypokalaemic periodic paralysis. To better define the functional consequences for a loss-of-function, we generated NaV1.4 null mice by deletion of exon 12. Heterozygous null mice have latent myasthenia and a right shift of the force-stimulus relation, without evidence of periodic paralysis. Sodium current density was half that of wild-type muscle and no compensation by retained expression of the foetal NaV1.5 isoform was detected. Mice null for NaV1.4 did not survive beyond the second postnatal day. This mouse model shows remarkable preservation of muscle function and viability for haploinsufficiency of NaV1.4, as has been reported in humans, with a propensity for pseudo-myasthenia caused by a marginal Na(+) current density to support sustained high-frequency action potentials in muscle.
Keywords: SCN4A; channelopathy; congenital myasthenic syndrome; skeletal muscle; weakness.
© The Author (2016). Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures


Similar articles
-
Hypokalaemic periodic paralysis and myotonia in a patient with homozygous mutation p.R1451L in NaV1.4.Sci Rep. 2018 Jun 26;8(1):9714. doi: 10.1038/s41598-018-27822-2. Sci Rep. 2018. PMID: 29946067 Free PMC article.
-
Myasthenic congenital myopathy from recessive mutations at a single residue in NaV1.4.Neurology. 2019 Mar 26;92(13):e1405-e1415. doi: 10.1212/WNL.0000000000007185. Epub 2019 Mar 1. Neurology. 2019. PMID: 30824560 Free PMC article.
-
Loss-of-function mutations in SCN4A cause severe foetal hypokinesia or 'classical' congenital myopathy.Brain. 2016 Mar;139(Pt 3):674-91. doi: 10.1093/brain/awv352. Epub 2015 Dec 22. Brain. 2016. PMID: 26700687 Free PMC article.
-
Sodium Channelopathies of Skeletal Muscle.Handb Exp Pharmacol. 2018;246:309-330. doi: 10.1007/164_2017_52. Handb Exp Pharmacol. 2018. PMID: 28939973 Free PMC article. Review.
-
New Challenges Resulting From the Loss of Function of Nav1.4 in Neuromuscular Diseases.Front Pharmacol. 2021 Oct 4;12:751095. doi: 10.3389/fphar.2021.751095. eCollection 2021. Front Pharmacol. 2021. PMID: 34671263 Free PMC article. Review.
Cited by
-
Hydrops fetalis due to loss of function of hNav1.4 channel via compound heterozygous variants.J Hum Genet. 2025 Jan;70(1):3-8. doi: 10.1038/s10038-024-01284-z. Epub 2024 Aug 21. J Hum Genet. 2025. PMID: 39164360 Free PMC article.
-
A204E mutation in Nav1.4 DIS3 exerts gain- and loss-of-function effects that lead to periodic paralysis combining hyper- with hypo-kalaemic signs.Sci Rep. 2018 Nov 12;8(1):16681. doi: 10.1038/s41598-018-34750-8. Sci Rep. 2018. PMID: 30420713 Free PMC article.
-
Hypokalaemic periodic paralysis and myotonia in a patient with homozygous mutation p.R1451L in NaV1.4.Sci Rep. 2018 Jun 26;8(1):9714. doi: 10.1038/s41598-018-27822-2. Sci Rep. 2018. PMID: 29946067 Free PMC article.
-
β2-Adrenergic receptor agonists ameliorate the adverse effect of long-term pyridostigmine on neuromuscular junction structure.Brain. 2019 Dec 1;142(12):3713-3727. doi: 10.1093/brain/awz322. Brain. 2019. PMID: 31633155 Free PMC article.
-
Myasthenic congenital myopathy from recessive mutations at a single residue in NaV1.4.Neurology. 2019 Mar 26;92(13):e1405-e1415. doi: 10.1212/WNL.0000000000007185. Epub 2019 Mar 1. Neurology. 2019. PMID: 30824560 Free PMC article.
References
-
- Burgess DL, Kohrman DC, Galt J, Plummer NW, Jones JM, Spear B, et al. Mutation of a new sodium channel gene, Scn8a, in the mouse mutant 'motor endplate disease'. Nat Genet 1995; 10: 461–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

