Necroptosis-independent signaling by the RIP kinases in inflammation
- PMID: 27048814
- PMCID: PMC4889460
- DOI: 10.1007/s00018-016-2203-4
Necroptosis-independent signaling by the RIP kinases in inflammation
Abstract
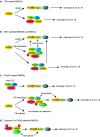
Recent advances have identified a signaling cascade involving receptor interacting protein kinase 1 (RIPK1), RIPK3 and the pseudokinase mixed lineage kinase domain-like (MLKL) that is crucial for induction of necroptosis, a non-apoptotic form of cell death. RIPK1-RIPK3-MLKL-mediated necroptosis has been attributed to cause many inflammatory diseases through the release of cellular damage-associated molecular patterns (DAMPs). In addition to necroptosis, emerging evidence suggests that these necroptosis signal adaptors can also facilitate inflammation independent of cell death. In particular, the RIP kinases can drive NF-κB and inflammasome activation independent of cell death. In this review, we will discuss recent discoveries that led to this realization and present arguments why cell death-independent signaling by the RIP kinases may have a more important role in inflammation than necroptosis.
Keywords: IL-1β; NF-κB; RIPK1; RIPK3; RelB.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

