Simultaneous blockade of VEGF and Dll4 by HD105, a bispecific antibody, inhibits tumor progression and angiogenesis
- PMID: 27049350
- PMCID: PMC4968104
- DOI: 10.1080/19420862.2016.1171432
Simultaneous blockade of VEGF and Dll4 by HD105, a bispecific antibody, inhibits tumor progression and angiogenesis
Abstract
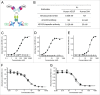
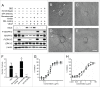
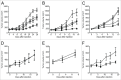
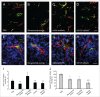
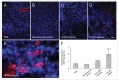
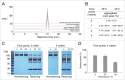
Several angiogenesis inhibitors targeting the vascular endothelial growth factor (VEGF) signaling pathway have been approved for cancer treatment. However, VEGF inhibitors alone were shown to promote tumor invasion and metastasis by increasing intratumoral hypoxia in some preclinical and clinical studies. Emerging reports suggest that Delta-like ligand 4 (Dll4) is a promising target of angiogenesis inhibition to augment the effects of VEGF inhibitors. To evaluate the effects of simultaneous blockade against VEGF and Dll4, we developed a bispecific antibody, HD105, targeting VEGF and Dll4. The HD105 bispecific antibody, which is composed of an anti-VEGF antibody (bevacizumab-similar) backbone C-terminally linked with a Dll4-targeting single-chain variable fragment, showed potent binding affinities against VEGF (KD: 1.3 nM) and Dll4 (KD: 30 nM). In addition, the HD105 bispecific antibody competitively inhibited the binding of ligands to their receptors, i.e., VEGF to VEGFR2 (EC50: 2.84 ± 0.41 nM) and Dll4 to Notch1 (EC50: 1.14 ± 0.06 nM). Using in vitro cell-based assays, we found that HD105 effectively blocked both the VEGF/VEGFR2 and Dll4/Notch1 signaling pathways in endothelial cells, resulting in a conspicuous inhibition of endothelial cell proliferation and sprouting. HD105 also suppressed Dll4-induced Notch1-dependent activation of the luciferase gene. In vivo xenograft studies demonstrated that HD105 more efficiently inhibited the tumor progression of human A549 lung and SCH gastric cancers than an anti-VEGF antibody or anti-Dll4 antibody alone. In conclusion, HD105 may be a novel therapeutic bispecific antibody for cancer treatment.
Keywords: Anti-angiogenesis; VEGF; anti-cancer; biologics; delta-like ligand; therapeutic antibody.
Figures
References
-
- Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J 1999; 13:9-22; PMID:9872925 - PubMed
-
- Meadows KL, Hurwitz HI. Anti-VEGF therapies in the clinic. Cold Spring Harb Perspect Med 2012; 2:a006577; PMID:23028128; http://dx.doi.org/10.1101/cshperspect.a006577 - DOI - PMC - PubMed
-
- Casak SJ, Fashoyin-Aje I, Lemery SJ, Zhang L, Jin R, Li H, Zhao L, Zhao H, Zhang H, Chen H, et al.. FDA Approval Summary: Ramucirumab for Gastric Cancer. Clin Cancer Res 2015; 21:3372-6; PMID:26048277; http://dx.doi.org/10.1158/1078-0432.CCR-15-0600 - DOI - PubMed
-
- Trichonas G, Kaiser PK. Aflibercept for the treatment of age-related macular degeneration. Ophthalmol Ther. 2013; 2:89-98; PMID:25135809; http://dx.doi.org/10.1007/s40123-013-0015-2 - DOI - PMC - PubMed
-
- Bergers G, Hanahan D. Modes of resistance to anti-angiogenic therapy. Nat Rev Cancer 2008; 8:592-603; PMID:18650835; http://dx.doi.org/10.1038/nrc2442 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
