Autoantibody-Positive Healthy Individuals Display Unique Immune Profiles That May Regulate Autoimmunity
- PMID: 27059145
- PMCID: PMC5042816
- DOI: 10.1002/art.39706
Autoantibody-Positive Healthy Individuals Display Unique Immune Profiles That May Regulate Autoimmunity
Abstract
Objective: Antinuclear antibodies (ANAs) are detected in ∼18% of females, yet autoimmune disease develops in only 5-8%. Immunologic differences between ANA-positive healthy individuals and patients with systemic lupus erythematosus (SLE) may elucidate the regulatory mechanisms by which ANA-positive individuals avoid transition to clinical autoimmune disease.
Methods: Healthy individuals (n = 790) were screened for autoantibodies specific for 11 antigens associated with lupus, systemic sclerosis, and Sjögren's syndrome. From this screening, 31 European American ANA-positive healthy individuals were selected and demographically matched to ANA-negative controls and SLE patients. Serum cytokine profiles, leukocyte subset frequency, and reactivity were analyzed by multiplex assays, immunophenotyping, and phosphospecific flow cytometry.
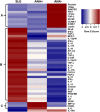
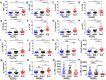
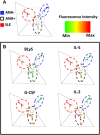
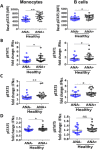
Results: Of 790 individuals screened, 57 (7%) were ANA-positive. The majority of proinflammatory cytokines, including interferon-γ (IFNγ), tumor necrosis factor, interleukin-17 (IL-17), and granulocyte colony-stimulating factor, exhibited a stepwise increase in serum levels from ANA-negative controls to ANA-positive healthy individuals to SLE patients (P < 0.0001). IFNα, IFNβ, IL-12p40, and stem cell factor/c-Kit ligand were increased in SLE patients only (P < 0.05). B lymphocyte stimulator (BlyS) was elevated in SLE patients but decreased in ANA-positive individuals (P < 0.001). Further, IL-1 receptor antagonist (IL-1Ra) was down-regulated in SLE patients only (P < 0.0001). ANA-positive individuals had increased frequencies of monocytes, memory B cells, and plasmablasts and increased levels of pSTAT-1 and pSTAT-3 following IFNα stimulation compared with ANA-negative controls (P < 0.05).
Conclusion: ANA-positive healthy individuals exhibit dysregulation in multiple immune pathways yet differ from SLE patients by the absence of elevated IFNs, BLyS, IL-12p40, and stem cell factor/c-Kit ligand. Further, severely decreased levels of IL-1Ra in SLE patients compared with ANA-positive individuals may contribute to disease development. These results highlight the importance of IFN-related pathways and regulatory elements in SLE pathogenesis.
© 2016 The Authors. Arthritis & Rheumatology published by Wiley Periodicals, Inc. on behalf of the American College of Rheumatology.
Conflict of interest statement
No conflicts of interest to report
Figures

Comment in
-
Editorial: Subduing Lupus: Can Preclinical Autoimmune Disease Be Arrested?Arthritis Rheumatol. 2016 Oct;68(10):2357-60. doi: 10.1002/art.39804. Arthritis Rheumatol. 2016. PMID: 27390125 Free PMC article. No abstract available.
References
-
- Heinlen LD, McClain MT, Merrill J, Akbarali YW, Edgerton CC, Harley JB, et al. Clinical criteria for systemic lupus erythematosus precede diagnosis, and associated autoantibodies are present before clinical symptoms. Arthritis Rheum 2007;56:2344–51. - PubMed
-
- Arbuckle MR, McClain MT, Rubertone MV, Scofield RH, Dennis GJ, James JA, et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N Engl J Med 2003;349:1526–33. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

