Valve-in-Valve Transcatheter Valve Implantation in the Nonaortic Position
- PMID: 27059552
- PMCID: PMC5142517
- DOI: 10.1111/jocs.12745
Valve-in-Valve Transcatheter Valve Implantation in the Nonaortic Position
Abstract
Background: Transcatheter valve-in-valve (VIV) procedures are an alternative to standard surgical valve replacement in high risk patients.
Methods: Cases in which a commercially approved transcatheter aortic valve replacement (TAVR) device was used for a nonaortic VIV procedure between November 2013 and September 2015 are reviewed. Clinical, echocardiographic, and procedural details, patient survival, and symptom severity by NYHA class at follow-up were assessed.
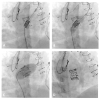
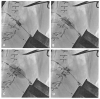
Results: All patients were heart-team determined high risk for conventional redo surgery (mean STS PROM = 6.8 ± 2.2%). Five patients underwent VIV replacement in the nonaortic position, four for bioprosthetic mitral valve dysfunction, and one for bioprosthetic tricuspid valve dysfunction. Bioprosthetic failure was due to stenosis in three patients and regurgitation in two others. A balloon-expandable device was used for all patients (Edwards Lifesciences, Irvine, CA, USA). Transcatheter VIV replacement was accomplished by the transapical (mitral) and transfemoral venous (tricuspid) approaches. Median postoperative length of stay was five days (range 3-12). No deaths occurred at a mean follow-up of 21 months. NYHA class at follow-up decreased from class IV at baseline to class I or II for all patients. No paravalvular leaks greater than trivial were encountered. Median mean gradient following mitral replacement was 6.5 mmHg (range 6-13 mmHg), and following tricuspid replacement was 4 mmHg. Postoperative complications included hematuria, epistaxis, acute kidney injury, and atrial fibrillation.
Conclusions: Transcatheter VIV implantation in the nonaortic position for dysfunctional bioprostheses can be performed safely with favorable clinical outcomes using a balloon expandable TAVR device. doi: 10.1111/jocs.12745 (J Card Surg 2016;31:282-288).
© 2016 Wiley Periodicals, Inc.
Figures



References
-
- Paradis J, Del Trigo M, Puri R. Transcatheter valve-in-valve and valve-in-ring for treating aortic and mitral surgical prosthetic dysfunction. J Am Coll Cardiol. 2015;66:2019–37. - PubMed
-
- Dvir D, Webb J, Brecker S, et al. Transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: results from the global valve-in-valve registry. Circulation. 2012;126(19):2335–44. - PubMed
-
- Gurvitch R, Cheung A, Ye J, et al. Transcatheter valve-in-valve implantation for failed surgical bioprosthetic valves. J Am Coll Cardiol. 2011;58(21):2196–2209. - PubMed
-
- Cheung A, Webb JG, Barbanti M, et al. 5-Year experience with transcatheter transapical mitral valve-in-valve implantation for bioprosthetic valve dysfunction. J Am Coll Cardiol. 2013;61:1759–66. - PubMed
-
- Cheung A, Webb JG, Wong DR, et al. Transapical transcatheter mitral valve-in-valve implantation in a human. Ann Thorac Surg. 2009;87:e18–20. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

