Gene therapy for immune tolerance induction in hemophilia with inhibitors
- PMID: 27061380
- PMCID: PMC4907803
- DOI: 10.1111/jth.13331
Gene therapy for immune tolerance induction in hemophilia with inhibitors
Abstract
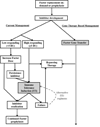
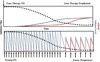
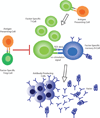
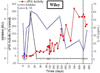
The development of inhibitors, i.e. neutralizing alloantibodies against factor (F) VIII or FIX, is the most significant complication of protein replacement therapy for patients with hemophilia, and is associated with both increased mortality and substantial physical, psychosocial and financial morbidity. Current management, including bypassing agents to treat and prevent bleeding, and immune tolerance induction for inhibitor eradication, is suboptimal for many patients. Fortunately, there are several emerging gene therapy approaches aimed at addressing these unmet clinical needs of patients with hemophilia and inhibitors. Herein, we review the mounting evidence from preclinical hemophilia models that the continuous uninterrupted expression of FVIII or FIX delivered as gene therapy can bias the immune system towards tolerance induction, and even promote the eradication of pre-existing inhibitors. We also discuss several gene transfer approaches that directly target immune cells in order to promote immune tolerance. These preclinical findings also shed light on the immunologic mechanisms that underlie tolerance induction.
Keywords: alloantibodies; factor IX; factor VIII; gene therapy; hemophilia; immune tolerance.
© 2016 International Society on Thrombosis and Haemostasis.
Figures

References
-
- DiMichele DM, Hoots WK, Pipe SW, Rivard GE, Santagostino E. International workshop on immune tolerance induction: consensus recommendations. Haemophilia. 2007;13(Suppl 1):1–22. - PubMed
-
- Kempton CL, Meeks SL. Toward optimal therapy for inhibitors in hemophilia. Blood. 2014;124:3365–3372. - PubMed
-
- DiMichele D. Inhibitor development in haemophilia B: an orphan disease in need of attention. British Journal of Haematology. 2007;138:305–315. - PubMed
-
- Walsh CE, Soucie JM, Miller CH United States Hemophilia Treatment Center N. Impact of inhibitors on hemophilia A mortality in the United States. Am J Hematol. 2015;90:400–405. - PubMed
-
- Eckhardt CL, Loomans JI, van Velzen AS, Peters M, Mauser-Bunschoten EP, Schwaab R, Mazzucconi MG, Tagliaferri A, Siegmund B, Reitter-Pfoertner SE, van der Bom JG, Fijnvandraat K Group IS. Inhibitor development and mortality in non-severe hemophilia A. J Thromb Haemost. 2015;13:1217–1225. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

