Production impact of influenza A(H1N1)pdm09 virus infection on fattening pigs in Norway
- PMID: 27065145
- PMCID: PMC7109966
- DOI: 10.2527/jas.2015-9251
Production impact of influenza A(H1N1)pdm09 virus infection on fattening pigs in Norway
Abstract
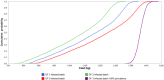
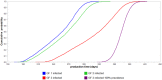
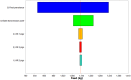
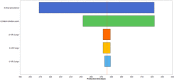
Newly emerged influenza A(H1N1)pdm09 virus infection in Norwegian pigs, although often observed in a subclinical form, can lower the pig's growth performance by reducing feed efficiency in terms of a poorer feed conversion ratio. Infected pigs would consume more feed and require protracted production time to reach market weight. In our observational longitudinal study, growth performance data from 728 control pigs and 193 infected pigs with known viral shedding time points were analyzed using mixed linear regression models to give estimates of the marginal effects of infection. Gaussian curves describing the variability of the estimates at the individual pig level formed the fundamental inputs to our stochastic models. The models were constructed to simulate the summed negative effects of the infection at the batch level of 150 fattening pigs growing from 33 to 100 kg. Other inputs of variability and uncertainty were 1) batch transmission points, 2) pig infection points to reflect the disease transmission dynamics of the virus, and 3) final prevalence of infected pigs in the batch. Monte Carlo random sampling gave 5,000 estimates on the outputs of the marginal effects for each pig. These results were summed up to provide estimates for a batch size of 150 pigs. This figure was adjusted by our final prevalence distribution function, which was also derived from the longitudinal study with 12 cohorts of infected pigs. For a 150-fattening-pig herd randomly selected from the population, the marginal effects of the infection were 1) 835 kg (fifth percentile) to 1,350 kg (95th percentile) increased feed intake and 2) 194 (fifth percentile) to 334 (95th percentile) pig days in excess of expected figures for an uninfected batch. A batch infected during growth phase 3 (81 to 100 kg BW) gave the worst results since the longitudinal study showed that a pig infected during growth phase 3 required more feed and a greater protracted production time compared to younger infected pigs. Sensitivity analysis showed that final prevalence had the greatest impact on the conditional mean and variation of the marginal effects of infections. Batch transmission point was the next most influential factor. Lowering the final prevalence and preventing older fattening pigs from being infected will have the greatest benefit in saving feed cost and reducing delay in getting the pigs to the market.
Figures
References
-
- Brookes S. M., Núñez A., Choudhury B., Matrosovich M., Essen S. C., Clifford D., Slomka M. J., Kuntz-Simon G., Garcon F., Nash B., Hanna A., Heegaard P. M. H., Quéguiner S., Chiapponi C., Bublot M., Maldonado Garcia J., Gardner R., Foni E., Loeffen W., Larsen L., Van Reeth K., Banks J., Irvine R. M., Brown I. H. 2010. Replication, pathogenesis and transmission of pandemic (H1N1) 2009 virus in non-immune pigs. PLoS ONE 5(2):e9068. doi:10.1371/journal.pone.0009068 - DOI - PMC - PubMed
-
- Brown I. H. 2000. The epidemiology and evolution of influenza viruses in pigs. Vet. Microbiol. 74 (1–2):29–46. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

