Intermittent Theta-Burst Transcranial Magnetic Stimulation Alters Electrical Properties of Fast-Spiking Neocortical Interneurons in an Age-Dependent Fashion
- PMID: 27065812
- PMCID: PMC4811908
- DOI: 10.3389/fncir.2016.00022
Intermittent Theta-Burst Transcranial Magnetic Stimulation Alters Electrical Properties of Fast-Spiking Neocortical Interneurons in an Age-Dependent Fashion
Abstract
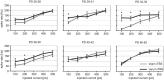
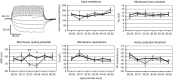
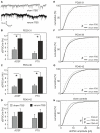
Modulation of human cortical excitability by repetitive transcranial magnetic stimulation (rTMS) appears to be in part related to changed activity of inhibitory systems. Our own studies showed that intermittent theta-burst stimulation (iTBS) applied via rTMS to rat cortex primarily affects the parvalbumin-expressing (PV) fast-spiking interneurons (FSIs), evident via a strongly reduced PV expression. We further found the iTBS effect on PV to be age-dependent since no reduction in PV could be induced before the perineuronal nets (PNNs) of FSIs start to grow around postnatal day (PD) 30. To elucidate possible iTBS-induced changes in the electrical properties of FSIs and cortical network activity during cortical critical period, we performed ex vivo-in vitro whole-cell patch clamp recordings from pre-labeled FSIs in the current study. FSIs of verum iTBS-treated rats displayed a higher excitability than sham-treated controls at PD29-38, evident as higher rates of induced action potential firing at low current injections (100-200 pA) and a more depolarized resting membrane potential. This effect was absent in younger (PD26-28) and older animals (PD40-62). Slices of verum iTBS-treated rats further showed higher rates of spontaneous excitatory postsynaptic currents (sEPSCs). Based on these and previous findings we conclude that FSIs are particularly sensitive to TBS during early cortical development, when FSIs show an activity-driven step of maturation which is paralleled by intense growth of the PNNs and subsequent closure of the cortical critical period. Although to be proven further, rTMS may be a possible early intervention to compensate for hypo-activity related mal-development of cortical neuronal circuits.
Keywords: calcium-binding protein; cortical fast spiking interneurons; development; parvalbumin; repetitive transcranial magnetic stimulation.
Figures


Similar articles
-
Reduction in cortical parvalbumin expression due to intermittent theta-burst stimulation correlates with maturation of the perineuronal nets in young rats.Dev Neurobiol. 2015 Jan;75(1):1-11. doi: 10.1002/dneu.22205. Epub 2014 Jun 26. Dev Neurobiol. 2015. PMID: 24962557
-
Multiple blocks of intermittent and continuous theta-burst stimulation applied via transcranial magnetic stimulation differently affect sensory responses in rat barrel cortex.J Physiol. 2015 Feb 15;593(4):967-85. doi: 10.1113/jphysiol.2014.282467. Epub 2015 Jan 12. J Physiol. 2015. PMID: 25504571 Free PMC article.
-
Neuropeptide Y as a possible homeostatic element for changes in cortical excitability induced by repetitive transcranial magnetic stimulation.Brain Stimul. 2018 Jul-Aug;11(4):797-805. doi: 10.1016/j.brs.2018.02.017. Epub 2018 Feb 24. Brain Stimul. 2018. PMID: 29519725
-
Modulation of cortical inhibition by rTMS - findings obtained from animal models.J Physiol. 2011 Sep 15;589(Pt 18):4423-35. doi: 10.1113/jphysiol.2011.206573. Epub 2011 Jul 18. J Physiol. 2011. PMID: 21768267 Free PMC article. Review.
-
Involvement of cortical fast-spiking parvalbumin-positive basket cells in epilepsy.Prog Brain Res. 2016;226:81-126. doi: 10.1016/bs.pbr.2016.04.012. Epub 2016 Jun 7. Prog Brain Res. 2016. PMID: 27323940 Free PMC article. Review.
Cited by
-
Parvalbumin Role in Epilepsy and Psychiatric Comorbidities: From Mechanism to Intervention.Front Integr Neurosci. 2022 Feb 17;16:765324. doi: 10.3389/fnint.2022.765324. eCollection 2022. Front Integr Neurosci. 2022. PMID: 35250498 Free PMC article. Review.
-
Low-intensity transcranial magnetic stimulation promotes the survival and maturation of newborn oligodendrocytes in the adult mouse brain.Glia. 2019 Aug;67(8):1462-1477. doi: 10.1002/glia.23620. Epub 2019 Apr 16. Glia. 2019. PMID: 30989733 Free PMC article.
-
Extensive Structural Remodeling of the Axonal Arbors of Parvalbumin Basket Cells during Development in Mouse Neocortex.J Neurosci. 2021 Nov 10;41(45):9326-9339. doi: 10.1523/JNEUROSCI.0871-21.2021. Epub 2021 Sep 28. J Neurosci. 2021. PMID: 34583957 Free PMC article.
-
Conduction Velocity Along the Local Axons of Parvalbumin Interneurons Correlates With the Degree of Axonal Myelination.Cereb Cortex. 2021 Jun 10;31(7):3374-3392. doi: 10.1093/cercor/bhab018. Cereb Cortex. 2021. PMID: 33704414 Free PMC article.
-
Mechanism of Repetitive Transcranial Magnetic Stimulation for Depression.Shanghai Arch Psychiatry. 2018 Apr 25;30(2):84-92. doi: 10.11919/j.issn.1002-0829.217047. Shanghai Arch Psychiatry. 2018. PMID: 29736128 Free PMC article. Review.
References
-
- Bashir S., Perez J. M., Horvath J. C., Pena-Gomez C., Vernet M., Capia A., et al. . (2014). Differential effects of motor cortical excitability and plasticity in young and old individuals: a transcranial magnetic stimulation (TMS) study. Front. Aging Neurosci. 6:111. 10.3389/fnagi.2014.00111 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

