SPAK and OSR1 play essential roles in potassium homeostasis through actions on the distal convoluted tubule
- PMID: 27068441
- PMCID: PMC5009767
- DOI: 10.1113/JP272311
SPAK and OSR1 play essential roles in potassium homeostasis through actions on the distal convoluted tubule
Abstract
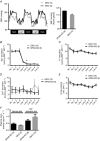
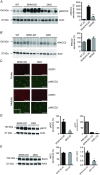
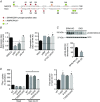
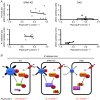
Key points: STE20 (Sterile 20)/SPS-1 related proline/alanine-rich kinase (SPAK) and oxidative stress-response kinase-1 (OSR1) phosphorylate and activate the renal Na(+) -K(+) -2Cl(-) cotransporter 2 (NKCC2) and Na(+) Cl(-) cotransporter (NCC). Mouse models suggest that OSR1 mainly activates NKCC2-mediated sodium transport along the thick ascending limb, while SPAK mainly activates NCC along the distal convoluted tubule, but the kinases may compensate for each other. We hypothesized that disruption of both kinases would lead to polyuria and severe salt-wasting, and generated SPAK/OSR1 double knockout mice to test this. Despite a lack of SPAK and OSR1, phosphorylated NKCC2 abundance was still high, suggesting the existence of an alternative activating kinase. Compensatory changes in SPAK/OSR1-independent phosphorylation sites on both NKCC2 and NCC and changes in sodium transport along the collecting duct were also observed. Potassium restriction revealed that SPAK and OSR1 play essential roles in the emerging model that NCC activation is central to sensing changes in plasma [K(+) ].
Abstract: STE20 (Sterile 20)/SPS-1 related proline/alanine-rich kinase (SPAK) and oxidative stress-response kinase-1 (OSR1) activate the renal cation cotransporters Na(+) -K(+) -2Cl(-) cotransporter (NKCC2) and Na(+) -Cl(-) cotransporter (NCC) via phosphorylation. Knockout mouse models suggest that OSR1 mainly activates NKCC2, while SPAK mainly activates NCC, with possible cross-compensation. We tested the hypothesis that disrupting both kinases causes severe polyuria and salt-wasting by generating SPAK/OSR1 double knockout (DKO) mice. DKO mice displayed lower systolic blood pressure compared with SPAK knockout (SPAK-KO) mice, but displayed no severe phenotype even after dietary salt restriction. Phosphorylation of NKCC2 at SPAK/OSR1-dependent sites was lower than in SPAK-KO mice, but still significantly greater than in wild type mice. In the renal medulla, there was significant phosphorylation of NKCC2 at SPAK/OSR1-dependent sites despite a complete absence of SPAK and OSR1, suggesting the existence of an alternative activating kinase. The distal convoluted tubule has been proposed to sense plasma [K(+) ], with NCC activation serving as the primary effector pathway that modulates K(+) secretion, by metering sodium delivery to the collecting duct. Abundance of phosphorylated NCC (pNCC) is dramatically lower in SPAK-KO mice than in wild type mice, and the additional disruption of OSR1 further reduced pNCC. SPAK-KO and kidney-specific OSR1 single knockout mice maintained plasma [K(+) ] following dietary potassium restriction, but DKO mice developed severe hypokalaemia. Unlike mice lacking SPAK or OSR1 alone, DKO mice displayed an inability to phosphorylate NCC under these conditions. These data suggest that SPAK and OSR1 are essential components of the effector pathway that maintains plasma [K(+) ].
© 2016 The Authors. The Journal of Physiology © 2016 The Physiological Society.
Figures



Similar articles
-
Impaired phosphorylation of Na(+)-K(+)-2Cl(-) cotransporter by oxidative stress-responsive kinase-1 deficiency manifests hypotension and Bartter-like syndrome.Proc Natl Acad Sci U S A. 2011 Oct 18;108(42):17538-43. doi: 10.1073/pnas.1107452108. Epub 2011 Oct 4. Proc Natl Acad Sci U S A. 2011. PMID: 21972418 Free PMC article.
-
SPAK isoforms and OSR1 regulate sodium-chloride co-transporters in a nephron-specific manner.J Biol Chem. 2012 Nov 2;287(45):37673-90. doi: 10.1074/jbc.M112.402800. Epub 2012 Sep 12. J Biol Chem. 2012. PMID: 22977235 Free PMC article.
-
Dysregulation of the WNK4-SPAK/OSR1 pathway has a minor effect on baseline NKCC2 phosphorylation.Am J Physiol Renal Physiol. 2024 Jan 1;326(1):F39-F56. doi: 10.1152/ajprenal.00100.2023. Epub 2023 Oct 26. Am J Physiol Renal Physiol. 2024. PMID: 37881876 Free PMC article.
-
Role of SPAK and OSR1 signalling in the regulation of NaCl cotransporters.Curr Opin Nephrol Hypertens. 2011 Sep;20(5):534-40. doi: 10.1097/MNH.0b013e3283484b06. Curr Opin Nephrol Hypertens. 2011. PMID: 21610494 Review.
-
Cotransporters, WNKs and hypertension: an update.Curr Opin Nephrol Hypertens. 2008 Mar;17(2):186-92. doi: 10.1097/MNH.0b013e3282f5244e. Curr Opin Nephrol Hypertens. 2008. PMID: 18277153 Review.
Cited by
-
Differential roles of WNK4 in regulation of NCC in vivo.Am J Physiol Renal Physiol. 2018 May 1;314(5):F999-F1007. doi: 10.1152/ajprenal.00177.2017. Epub 2018 Jan 31. Am J Physiol Renal Physiol. 2018. PMID: 29384416 Free PMC article.
-
The WNK signaling pathway and salt-sensitive hypertension.Hypertens Res. 2020 Aug;43(8):733-743. doi: 10.1038/s41440-020-0437-x. Epub 2020 Apr 14. Hypertens Res. 2020. PMID: 32286498 Review.
-
WNKs are potassium-sensitive kinases.Am J Physiol Cell Physiol. 2021 May 1;320(5):C703-C721. doi: 10.1152/ajpcell.00456.2020. Epub 2021 Jan 13. Am J Physiol Cell Physiol. 2021. PMID: 33439774 Free PMC article.
-
WNK4 kinase: from structure to physiology.Am J Physiol Renal Physiol. 2021 Mar 1;320(3):F378-F403. doi: 10.1152/ajprenal.00634.2020. Epub 2021 Jan 25. Am J Physiol Renal Physiol. 2021. PMID: 33491560 Free PMC article. Review.
-
Novel CUL3 Variant Causing Familial Hyperkalemic Hypertension Impairs Regulation and Function of Ubiquitin Ligase Activity.Hypertension. 2022 Jan;79(1):60-75. doi: 10.1161/HYPERTENSIONAHA.121.17624. Epub 2021 Dec 2. Hypertension. 2022. PMID: 34878901 Free PMC article.
References
-
- Bazua‐Valenti S, Chavez‐Canales M, Rojas‐Vega L, Gonzalez‐Rodriguez X, Vazquez N, Rodriguez‐Gama A, Argaiz ER, Melo Z, Plata C, Ellison DH, Garcia‐Valdes J, Hadchouel J & Gamba G (2015). The Effect of WNK4 on the Na+–Cl– cotransporter is modulated by intracellular chloride. J Am Soc Nephrol 26, 1781–1786. - PMC - PubMed
-
- Bhalla V & Hallows KR (2008). Mechanisms of ENaC regulation and clinical implications. J Am Soc Nephrol 19, 1845–1854. - PubMed
-
- Boersema PJ, Raijmakers R, Lemeer S, Mohammed S & Heck AJ (2009). Multiplex peptide stable isotope dimethyl labeling for quantitative proteomics. Nat Protoc 4, 484–494. - PubMed
-
- Bostanjoglo M, Reeves WB, Reilly RF, Velazquez H, Robertson N, Litwack G, Morsing P, Dorup J, Bachmann S & Ellison DH (1998). 11β‐hydroxysteroid dehydrogenase, mineralocorticoid receptor, and thiazide‐sensitive Na–Cl cotransporter expression by distal tubules. J Am Soc Nephrol 9, 1347–1358. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

