Induction of Autophagy in the Striatum and Hypothalamus of Mice after 835 MHz Radiofrequency Exposure
- PMID: 27073885
- PMCID: PMC4830612
- DOI: 10.1371/journal.pone.0153308
Induction of Autophagy in the Striatum and Hypothalamus of Mice after 835 MHz Radiofrequency Exposure
Abstract
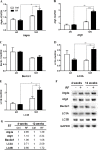
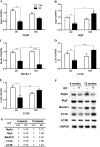
The extensive use of wireless mobile phones and associated communication devices has led to increasing public concern about potential biological health-related effects of the exposure to electromagnetic fields (EMFs). EMFs emitted by a mobile phone have been suggested to influence neuronal functions in the brain and affect behavior. However, the affects and phenotype of EMFs exposure are unclear. We applied radiofrequency (RF) of 835 MHz at a specific absorption rate (SAR) of 4.0 W/kg for 5 hours/day for 4 and 12 weeks to clarify the biological effects on mouse brain. Interestingly, microarray data indicated that a variety of autophagic related genes showed fold-change within small range after 835 MHz RF exposure. qRT-PCR revealed significant up-regulation of the autophagic genes Atg5, LC3A and LC3B in the striatum and hypothalamus after a 12-week RF. In parallel, protein expression of LC3B-II was also increased in both brain regions. Autophagosomes were observed in the striatum and hypothalamus of RF-exposed mice, based on neuronal transmission electron microscopy. Taken together, the results indicate that RF exposure of the brain can induce autophagy in neuronal tissues, providing insight into the protective mechanism or adaptation to RF stress.
Conflict of interest statement
Figures


References
-
- Dubreuil D, Jay T, Edeline J. Does head-only exposure to GSM-900 electromagnetic fields affect. Behav Brain Res. 2002;129:203–10. - PubMed
-
- Leszczynski D, Joenväärä S, Reivinen J, Kuokka R. Non-thermal activation of the hsp27/p38MAPK stress pathway by mobile phone radiation in human endothelial cells: Molecular mechanism for cancer- and blood-brain barrier-related effects. Differentiation. 2002;70(2–3):120–9. . - PubMed
-
- Nittby H, Brun A, Eberhardt J, Malmgren L, Persson BR, Salford LG. Increased blood-brain barrier permeability in mammalian brain 7 days after exposure to the radiation from a GSM-900 mobile phone. Pathophysiology: the official journal of the International Society for Pathophysiology / ISP. 2009;16(2–3):103–12. 10.1016/j.pathophys.2009.01.001 . - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

