Inter-polysomal coupling of termination and initiation during translation in eukaryotic cell-free system
- PMID: 27075299
- PMCID: PMC4830951
- DOI: 10.1038/srep24518
Inter-polysomal coupling of termination and initiation during translation in eukaryotic cell-free system
Abstract
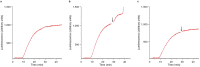
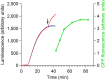
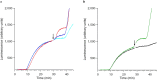
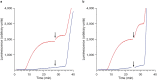
The recording of the luciferase-generated luminescence in the eukaryotic cell-free translation system programmed with mRNA encoding firefly luciferase (Luc-mRNA) showed that the addition of free exogenous mRNAs into the translation reactor induces the immediate release of the functionally active protein from the polyribosomes of the translation system. The phenomenon did not depend on the coding specificity of the added free mRNA. At the same time it depended on the "initiation potential" of the added mRNA (including the features that ensure the successful initiation of translation, such as the presence of the cap structure and the sufficient concentration of the added mRNA in the translation mixture). The phenomenon also strictly depended on the presence of the stop codon in the translated mRNA. As the above-mentioned features of the added mRNA imply its activity in initiation of a new translation, the experimental data are found in agreement with the scenario where the molecules of the added mRNA interact by their 5'-ends with terminating and recycling ribosomes, stimulating the release of the complete polypeptides and providing for the initiation of a new translation.
Figures
Similar articles
-
Translation of non-capped mRNAs in a eukaryotic cell-free system: acceleration of initiation rate in the course of polysome formation.Nucleic Acids Res. 2007;35(19):6547-59. doi: 10.1093/nar/gkm725. Epub 2007 Sep 26. Nucleic Acids Res. 2007. PMID: 17897963 Free PMC article.
-
Coupling of Translation Initiation and Termination Does Not Depend on the Mode of Initiation.Biochemistry (Mosc). 2017 Jul;82(7):816-820. doi: 10.1134/S0006297917070069. Biochemistry (Mosc). 2017. PMID: 28918745
-
Dissecting eukaryotic translation and its control by ribosome density mapping.Nucleic Acids Res. 2005 Apr 28;33(8):2421-32. doi: 10.1093/nar/gki331. Print 2005. Nucleic Acids Res. 2005. PMID: 15860778 Free PMC article.
-
Alternative translation start sites and hidden coding potential of eukaryotic mRNAs.Bioessays. 2008 Jul;30(7):683-91. doi: 10.1002/bies.20771. Bioessays. 2008. PMID: 18536038 Review.
-
[Termination step of protein biosynthesis and release of ribosomes from mRNA].Tanpakushitsu Kakusan Koso. 1972 Dec;17(12):897-905. Tanpakushitsu Kakusan Koso. 1972. PMID: 4569224 Review. Japanese. No abstract available.
Cited by
-
Diversity and Similarity of Termination and Ribosome Rescue in Bacterial, Mitochondrial, and Cytoplasmic Translation.Biochemistry (Mosc). 2021 Sep;86(9):1107-1121. doi: 10.1134/S0006297921090066. Biochemistry (Mosc). 2021. PMID: 34565314 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

