A vacuole-like compartment concentrates a disordered calcium phase in a key coccolithophorid alga
- PMID: 27075521
- PMCID: PMC4834641
- DOI: 10.1038/ncomms11228
A vacuole-like compartment concentrates a disordered calcium phase in a key coccolithophorid alga
Abstract
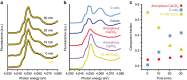
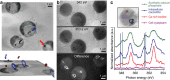
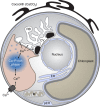
Coccoliths are calcitic particles produced inside the cells of unicellular marine algae known as coccolithophores. They are abundant components of sea-floor carbonates, and the stoichiometry of calcium to other elements in fossil coccoliths is widely used to infer past environmental conditions. Here we study cryo-preserved cells of the dominant coccolithophore Emiliania huxleyi using state-of-the-art nanoscale imaging and spectroscopy. We identify a compartment, distinct from the coccolith-producing compartment, filled with high concentrations of a disordered form of calcium. Co-localized with calcium are high concentrations of phosphorus and minor concentrations of other cations. The amounts of calcium stored in this reservoir seem to be dynamic and at a certain stage the compartment is in direct contact with the coccolith-producing vesicle, suggesting an active role in coccolith formation. Our findings provide insights into calcium accumulation in this important calcifying organism.
Figures




References
-
- Sikes C. S., Roer R. D. & Wilbur K. M. Photosynthesis and coccolith formation: Inorganic carbon sources and net inorganic reaction of deposition. Limnol. Oceanogr. 25, 248–261 (1980).
-
- Iglesias-Rodriguez M. D. et al.. Phytoplankton calcification in a high-CO2 world. Science 320, 336–340 (2008). - PubMed
-
- Beaufort L. et al.. Sensitivity of coccolithophores to carbonate chemistry and ocean acidification. Nature 476, 80–83 (2011). - PubMed
-
- Stoll H. M., Ziveri P. in Coccolithophores eds Thierstein H. R., Young J. R. Ch. 20, 529–562Springer (2004).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

