Therapeutic hypothermia attenuates tissue damage and cytokine expression after traumatic brain injury by inhibiting necroptosis in the rat
- PMID: 27080932
- PMCID: PMC4832230
- DOI: 10.1038/srep24547
Therapeutic hypothermia attenuates tissue damage and cytokine expression after traumatic brain injury by inhibiting necroptosis in the rat
Abstract
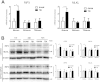
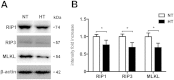
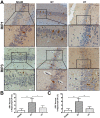
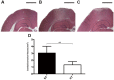
Necroptosis has been shown as an alternative form of cell death in many diseases, but the detailed mechanisms of the neuron loss after traumatic brain injury (TBI) in rodents remain unclear. To investigate whether necroptosis is induced after TBI and gets involved in the neuroprotecton of therapeutic hypothermia on the TBI, we observed the pathological and biochemical change of the necroptosis in the fluid percussion brain injury (FPI) model of the rats. We found that receptor-interacting protein (RIP) 1 and 3, and mixed lineage kinase domain-like protein (MLKL), the critical downstream mediators of necroptosis recently identified in vivo, as well as HMGB1 and the pro-inflammation cytokines TNF-α, IL-6 and IL-18, were increased at an early phase (6 h) in cortex after TBI. Posttraumatic hypothermia (33 °C) led to the decreases in the necroptosis regulators, inflammatory factors and brain tissue damage in rats compared with normothermia-treated TBI animals. Immunohistochemistry studies showed that posttraumatic hypothermia also decreased the necroptosis-associated proteins staining in injured cortex and hippocampal CA1. Therefore, we conclude that the RIP1/RIP3-MLKL-mediated necroptosis occurs after experimental TBI and therapeutic hypothermia may protect the injured central nervous system from tissue damage and the inflammatory responses by targeting the necroptosis signaling after TBI.
Figures

Similar articles
-
Mild Hypothermia Promotes Pericontusion Neuronal Sprouting via Suppressing Suppressor of Cytokine Signaling 3 Expression after Moderate Traumatic Brain Injury.J Neurotrauma. 2017 Apr 15;34(8):1636-1644. doi: 10.1089/neu.2016.4759. Epub 2017 Jan 13. J Neurotrauma. 2017. PMID: 27923323
-
Moderate hypothermia inhibits microglial activation after traumatic brain injury by modulating autophagy/apoptosis and the MyD88-dependent TLR4 signaling pathway.J Neuroinflammation. 2018 Sep 20;15(1):273. doi: 10.1186/s12974-018-1315-1. J Neuroinflammation. 2018. PMID: 30236124 Free PMC article.
-
Protective effect of mild-induced hypothermia against moderate traumatic brain injury in rats involved in necroptotic and apoptotic pathways.Brain Inj. 2017;31(3):406-415. doi: 10.1080/02699052.2016.1225984. Epub 2017 Jan 31. Brain Inj. 2017. PMID: 28140659
-
RIP3 in Necroptosis: Underlying Contributions to Traumatic Brain Injury.Neurochem Res. 2024 Feb;49(2):245-257. doi: 10.1007/s11064-023-04038-z. Epub 2023 Sep 25. Neurochem Res. 2024. PMID: 37743445 Review.
-
Necroptosis: Pathway diversity and characteristics.Semin Cell Dev Biol. 2015 Mar;39:56-62. doi: 10.1016/j.semcdb.2015.02.002. Epub 2015 Feb 13. Semin Cell Dev Biol. 2015. PMID: 25683283 Review.
Cited by
-
Translational Block in Stroke: A Constructive and "Out-of-the-Box" Reappraisal.Front Neurosci. 2021 May 14;15:652403. doi: 10.3389/fnins.2021.652403. eCollection 2021. Front Neurosci. 2021. PMID: 34054413 Free PMC article.
-
Combined Cyclosporin A and Hypothermia Treatment Inhibits Activation of BV-2 Microglia but Induces an Inflammatory Response in an Ischemia/Reperfusion Hippocampal Slice Culture Model.Front Cell Neurosci. 2019 Jun 25;13:273. doi: 10.3389/fncel.2019.00273. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31293389 Free PMC article.
-
The Role of Smurf1 in Neuronal Necroptosis after Lipopolysaccharide-Induced Neuroinflammation.Cell Mol Neurobiol. 2018 May;38(4):809-816. doi: 10.1007/s10571-017-0553-6. Epub 2017 Sep 22. Cell Mol Neurobiol. 2018. PMID: 28940129 Free PMC article.
-
Ginkgolide A attenuated apoptosis via inhibition of oxidative stress in mice with traumatic brain injury.Heliyon. 2024 Jan 14;10(2):e24759. doi: 10.1016/j.heliyon.2024.e24759. eCollection 2024 Jan 30. Heliyon. 2024. PMID: 38304806 Free PMC article.
-
Emerging Trends in the Use of Therapeutic Hypothermia as a Method for Neuroprotection in Brain Damage (Review).Sovrem Tekhnologii Med. 2021;12(5):94-104. doi: 10.17691/stm2020.12.5.11. Epub 2020 Oct 28. Sovrem Tekhnologii Med. 2021. PMID: 34796010 Free PMC article. Review.
References
-
- Lai Y. et al. Autophagy is increased after traumatic brain injury in mice and is partially inhibited by the antioxidant gamma-glutamylcysteinyl ethyl ester. J Cereb Blood Flow Metab. 28, 540–550 (2008). - PubMed
-
- Degterev A. et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 1, 112–119 (2005). - PubMed
-
- Pasparakis M. & Vandenabeele P. Necroptosis and its role in inflammation. Nature. 517, 311–320 (2015). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

