Developing a pro-regenerative biomaterial scaffold microenvironment requires T helper 2 cells
- PMID: 27081073
- PMCID: PMC4866509
- DOI: 10.1126/science.aad9272
Developing a pro-regenerative biomaterial scaffold microenvironment requires T helper 2 cells
Abstract
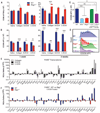
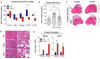
Immune-mediated tissue regeneration driven by a biomaterial scaffold is emerging as an innovative regenerative strategy to repair damaged tissues. We investigated how biomaterial scaffolds shape the immune microenvironment in traumatic muscle wounds to improve tissue regeneration. The scaffolds induced a pro-regenerative response, characterized by an mTOR/Rictor-dependent T helper 2 pathway that guides interleukin-4-dependent macrophage polarization, which is critical for functional muscle recovery. Manipulating the adaptive immune system using biomaterials engineering may support the development of therapies that promote both systemic and local pro-regenerative immune responses, ultimately stimulating tissue repair.
Copyright © 2016, American Association for the Advancement of Science.
Figures


Comment in
-
TISSUE REGENERATION. A scaffold immune microenvironment.Science. 2016 Apr 15;352(6283):298. doi: 10.1126/science.aaf7587. Science. 2016. PMID: 27081059 No abstract available.
-
Tissue repair: Biological scaffolds modulate immune cells.Nat Rev Immunol. 2016 Apr 28;16(5):276-7. doi: 10.1038/nri.2016.52. Nat Rev Immunol. 2016. PMID: 27121649 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

