Architecture of the symmetric core of the nuclear pore
- PMID: 27081075
- PMCID: PMC5207208
- DOI: 10.1126/science.aaf1015
Architecture of the symmetric core of the nuclear pore
Abstract
The nuclear pore complex (NPC) controls the transport of macromolecules between the nucleus and cytoplasm, but its molecular architecture has thus far remained poorly defined. We biochemically reconstituted NPC core protomers and elucidated the underlying protein-protein interaction network. Flexible linker sequences, rather than interactions between the structured core scaffold nucleoporins, mediate the assembly of the inner ring complex and its attachment to the NPC coat. X-ray crystallographic analysis of these scaffold nucleoporins revealed the molecular details of their interactions with the flexible linker sequences and enabled construction of full-length atomic structures. By docking these structures into the cryoelectron tomographic reconstruction of the intact human NPC and validating their placement with our nucleoporin interactome, we built a composite structure of the NPC symmetric core that contains ~320,000 residues and accounts for ~56 megadaltons of the NPC's structured mass. Our approach provides a paradigm for the structure determination of similarly complex macromolecular assemblies.
Copyright © 2016, American Association for the Advancement of Science.
Conflict of interest statement
The authors declare no financial conflicts of interest.
Figures



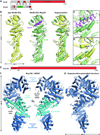
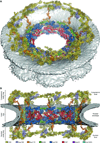


Comment in
-
Boundary Issues.Cell. 2016 May 5;165(4):759-61. doi: 10.1016/j.cell.2016.04.054. Cell. 2016. PMID: 27153485 No abstract available.
References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

