Combined Effects of High-Dose Bisphenol A and Oxidizing Agent (KBrO3) on Cellular Microenvironment, Gene Expression, and Chromatin Structure of Ku70-deficient Mouse Embryonic Fibroblasts
- PMID: 27082013
- PMCID: PMC4977032
- DOI: 10.1289/EHP237
Combined Effects of High-Dose Bisphenol A and Oxidizing Agent (KBrO3) on Cellular Microenvironment, Gene Expression, and Chromatin Structure of Ku70-deficient Mouse Embryonic Fibroblasts
Abstract
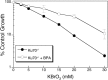
Background: Exposure to bisphenol A (BPA) has been reported to alter global gene expression, induce epigenetic modifications, and interfere with complex regulatory networks of cells. In addition to these reprogramming events, we have demonstrated that BPA exposure generates reactive oxygen species and promotes cellular survival when co-exposed with the oxidizing agent potassium bromate (KBrO3).
Objectives: We determined the cellular microenvironment changes induced by co-exposure of BPA and KBrO3 versus either agent alone.
Methods: Ku70-deficient cells were exposed to 150 μM BPA, 20 mM KBrO3, or co-exposed to both agents. Four and 24 hr post-damage initiation by KBrO3, with BPA-only samples timed to coincide with these designated time points, we performed whole-genome microarray analysis and evaluated chromatin structure, DNA lesion load, glutathione content, and intracellular pH.
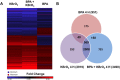
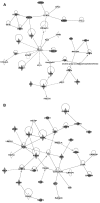
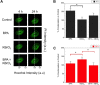
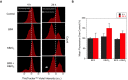
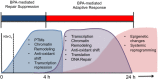
Results: We found that 4 hr post-damage initiation, BPA exposure and co-exposure transiently condensed chromatin compared with untreated and KBrO3-only treated cells; the transcription of DNA repair proteins was also reduced. At this time point, BPA exposure and co-exposure also reduced the change in intracellular pH observed after treatment with KBrO3 alone. Twenty-four hours post-damage initiation, BPA-exposed cells showed less condensed chromatin than cells treated with KBrO3 alone; the intracellular pH of the co-exposed cells was significantly reduced compared with untreated and KBrO3-treated cells; and significant up-regulation of DNA repair proteins was observed after co-exposure.
Conclusion: These results support the induction of an adaptive response by BPA co-exposure that alters the microcellular environment and modulates DNA repair. Further work is required to determine whether BPA induces similar DNA lesions in vivo at environmentally relevant doses; however, in the Ku70-deficient mouse embryonic fibroblasts, exposure to a high dose of BPA was associated with changes in the cellular microenvironment that may promote survival.
Citation: Gassman NR, Coskun E, Jaruga P, Dizdaroglu M, Wilson SH. 2016. Combined effects of high-dose bisphenol A and oxidizing agent (KBrO3) on cellular microenvironment, gene expression, and chromatin structure of Ku70-deficient mouse embryonic fibroblasts. Environ Health Perspect 124:1241-1252; http://dx.doi.org/10.1289/EHP237.
Conflict of interest statement
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Certain commercial equipment or materials are identified in this paper in order to specify adequately the experimental procedure. Such identification does not imply recommendation or endorsement by the National Institute of Standards and Technology, nor does it imply that the materials or equipment identified are necessarily the best available for the purpose.
The authors declare they have no actual or potential competing financial interests.
Figures

Similar articles
-
Bisphenol a promotes cell survival following oxidative DNA damage in mouse fibroblasts.PLoS One. 2015 Feb 18;10(2):e0118819. doi: 10.1371/journal.pone.0118819. eCollection 2015. PLoS One. 2015. PMID: 25693136 Free PMC article.
-
Epigenetic changes in p21 expression in renal cells after exposure to bromate.Toxicol Sci. 2014 Oct;141(2):432-40. doi: 10.1093/toxsci/kfu138. Epub 2014 Jul 11. Toxicol Sci. 2014. PMID: 25015661 Free PMC article.
-
Role of TET Dioxygenases and DNA Hydroxymethylation in Bisphenols-Stimulated Proliferation of Breast Cancer Cells.Environ Health Perspect. 2020 Feb;128(2):27008. doi: 10.1289/EHP5862. Epub 2020 Feb 27. Environ Health Perspect. 2020. PMID: 32105160 Free PMC article.
-
Oxidative DNA damage induced by potassium bromate under cell-free conditions and in mammalian cells.Carcinogenesis. 1995 Feb;16(2):335-42. doi: 10.1093/carcin/16.2.335. Carcinogenesis. 1995. PMID: 7859366
-
Toxicity and carcinogenicity of potassium bromate--a new renal carcinogen.Environ Health Perspect. 1990 Jul;87:309-35. doi: 10.1289/ehp.9087309. Environ Health Perspect. 1990. PMID: 2269236 Free PMC article. Review.
Cited by
-
High-Content Analysis Provides Mechanistic Insights into the Testicular Toxicity of Bisphenol A and Selected Analogues in Mouse Spermatogonial Cells.Toxicol Sci. 2017 Jan;155(1):43-60. doi: 10.1093/toxsci/kfw178. Epub 2016 Sep 14. Toxicol Sci. 2017. PMID: 27633978 Free PMC article.
-
Cardiovascular and renal oxidative stress-mediated toxicities associated with bisphenol-A exposures are mitigated by Curcuma longa in rats.Avicenna J Phytomed. 2024 Mar-Apr;14(2):202-214. doi: 10.22038/AJP.2023.23367. Avicenna J Phytomed. 2024. PMID: 38966628 Free PMC article.
-
Palmitate exacerbates bisphenol A toxicity via induction of ER stress and mitochondrial dysfunction.Biochim Biophys Acta Mol Cell Biol Lipids. 2021 Jan;1866(1):158816. doi: 10.1016/j.bbalip.2020.158816. Epub 2020 Sep 22. Biochim Biophys Acta Mol Cell Biol Lipids. 2021. PMID: 32976987 Free PMC article.
-
Oxidative Stress and BPA Toxicity: An Antioxidant Approach for Male and Female Reproductive Dysfunction.Antioxidants (Basel). 2020 May 10;9(5):405. doi: 10.3390/antiox9050405. Antioxidants (Basel). 2020. PMID: 32397641 Free PMC article. Review.
-
Effects of Bauhinia forficata on glycaemia, lipid profile, hepatic glycogen content and oxidative stress in rats exposed to Bisphenol A.Toxicol Rep. 2019 Mar 2;6:244-252. doi: 10.1016/j.toxrep.2019.03.001. eCollection 2019. Toxicol Rep. 2019. PMID: 30911467 Free PMC article.
References
-
- Adler S, Fraley DS. Potassium and intracellular pH. Kidney Int. 1977;11:433–442. - PubMed
-
- Babu S, Uppu S, Claville MO, Uppu RM. Prooxidant actions of bisphenol A (BPA) phenoxyl radicals: implications to BPA-related oxidative stress and toxicity. Toxicol Mech Methods. 2013;23:273–280. - PubMed
-
- Ballmaier D, Epe B. DNA damage by bromate: mechanism and consequences. Toxicology. 2006;221:166–171. - PubMed
-
- Belloc F, Dumain P, Boisseau MR, Jalloustre C, Reiffers J, Bernard P, et al. A flow cytometric method using Hoechst 33342 and propidium iodide for simultaneous cell cycle analysis and apoptosis determination in unfixed cells. Cytometry. 1994;17:59–65. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

