De Novo Sequencing of Complex Mixtures of Heparan Sulfate Oligosaccharides
- PMID: 27087275
- PMCID: PMC5068567
- DOI: 10.1021/acs.analchem.6b00519
De Novo Sequencing of Complex Mixtures of Heparan Sulfate Oligosaccharides
Abstract
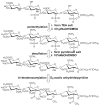
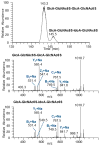
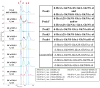
Here, we describe the first sequencing method of a complex mixture of heparan sulfate tetrasaccharides by LC-MS/MS. Heparin and heparan sulfate (HS) are linear polysaccharides that are modified in a complex manner by N- and O-sulfation, N-acetylation, and epimerization of the uronic acid. Heparin and HS are involved in various essential cellular communication processes. The structural analysis of these glycosaminoglycans is challenging due to the lability of their sulfate groups, the high heterogeneity of modifications, and the epimerization of the uronic acids. While advances in liquid chromatography (LC) and mass spectrometry (MS) have enabled compositional profiling of HS oligosaccharide mixtures, online separation and detailed structural analysis of isomeric and epimeric HS mixtures has not been achieved. Here, we report the development and evaluation of a chemical derivatization and tandem mass spectrometry method that can separate and identify isomeric and epimeric structures from complex mixtures. A series of well-defined synthetic HS tetrasaccharides varying in sulfation patterns and uronic acid epimerization were analyzed by chemical derivatization and LC-MS/MS. These synthetic compounds made it possible to establish relationships between HS structure, chromatographic behavior and MS/MS fragmentation characteristics. Using the analytical characteristics determined through the analysis of the synthetic HS tetrasaccharide standards, an HS tetrasacharide mixture derived from natural sources was successfully sequenced. This method represents the first sequencing of complex mixtures of HS oligosaccharides, an essential milestone in the analysis of structure-function relationships of these carbohydrates.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
-
- Gandhi NS, Mancera RL. Chem Biol Drug Des. 2008;72:455–482. - PubMed
-
- Rabenstein DL. Nat Prod Rep. 2002;19:312–331. - PubMed
-
- Tumova S, Woods A, Couchman JR. Int J Biochem Cell Biol. 2000;32:269–288. - PubMed
-
- Kresse H, Schonherr E. J Cell Physiol. 2001;189:266–274. - PubMed
-
- Fannon M, Forsten KE, Nugent MA. Biochemistry. 2000;39:1434–1445. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

