Validation of commercially available ELISAs for the detection of circulating sclerostin in hemodialysis patients
- PMID: 27088126
- PMCID: PMC4829955
- DOI: 10.15190/d.2016.2
Validation of commercially available ELISAs for the detection of circulating sclerostin in hemodialysis patients
Abstract
Background: Sclerostin is an endocrine regulator in chronic kidney disease - mineral and bone disorder (CKD-MBD). Validation of assay comparability and pre-analytical handling is mandatory for establishment of sclerostin as a biomarker.
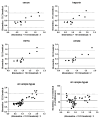
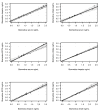
Methods: Blood samples (serum, EDTA, heparin and citrate plasma) were obtained from 12 hemodialysis (HD) patients after the long dialysis interval. Passing-Bablok regression analysis and Bland-Altman difference plots were used to evaluate the agreement between sclerostin levels measured with two commercially available ELISAs from TECOmedical and Biomedica.
Results: Independent of the sample type, the agreement of the two assays was poor with a strong proportional but no systematic bias. Compared to the TECOmedical assay, the Biomedica test yielded almost 2-fold higher sclerostin values throughout all sample types. Spike recovery and linear dilution studies revealed a higher accuracy of the TECOmedical assay (97% and 96%) compared to the Biomedica assay (118% and 78%). Sclerostin levels were stable within 4 hours after sample collection, in particular when analyzed in plasma. In contrast to the Biomedica assay, the TECOmedical showed a systematic but no proportional bias between serum and plasma samples with higher values for plasma samples. Among the 3 different plasma samples no systematic error could be documented.
Conclusion: Careful consideration of the pre-analytical handling and comparative assay validation are necessary to facilitate a more differentiated interpretation of studies reporting circulating sclerostin levels. The presence of a proportional bias demonstrates that in HD patients the two ELISAs for measuring sclerostin should not be used interchangeably. Furthermore, caution is necessary when comparing sclerostin results obtained from different blood sample types.
Keywords: CKD-MBD; biomarker; haemodialysis; sclerostin; vascular calcification.
Conflict of interest statement
M.H. is an employee of TECOmedical. The other authors declare to have no conflict of interest with respect to the present manuscript.
Figures



References
-
- Moe S, Drueke T, Cunningham J, et al. Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO) Kidney Int. 2006;69:1945–1953. - PubMed
-
- Brandenburg VM, D’Haese P, Deck A, et al. From skeletal to cardiovascular disease in 12 steps-the evolution of sclerostin as a major player in CKD-MBD. Pediatr Nephrol. 2016;31:195–206. - PubMed
-
- Sabbagh Y, Graciolli FG, O’Brien S, et al. Repression of osteocyte Wnt/beta-catenin signaling is an early event in the progression of renal osteodystrophy. J Bone Miner Res. 2012;27:1757–1772. - PubMed
-
- Ishimura E, Okuno S, Ichii M, et al. Relationship between serum sclerostin, bone metabolism markers, and bone mineral density in maintenance hemodialysis patients. J Clin Endocrinol Metab. 2014;99:4315–4320. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
