Subtypes of HPV-Positive Head and Neck Cancers Are Associated with HPV Characteristics, Copy Number Alterations, PIK3CA Mutation, and Pathway Signatures
- PMID: 27091409
- PMCID: PMC5026546
- DOI: 10.1158/1078-0432.CCR-16-0323
Subtypes of HPV-Positive Head and Neck Cancers Are Associated with HPV Characteristics, Copy Number Alterations, PIK3CA Mutation, and Pathway Signatures
Abstract
Purpose: There is substantial heterogeneity within human papillomavirus (HPV)-associated head and neck cancer (HNC) tumors that predispose them to different outcomes; however, the molecular heterogeneity in this subgroup is poorly characterized due to various historical reasons.
Experimental design: We performed unsupervised gene expression clustering on deeply annotated (transcriptome and genome) HPV(+) HNC samples from two cohorts (84 total primary tumors), including 18 HPV(-) HNC samples, to discover subtypes and characterize the differences between subgroups in terms of their HPV characteristics, pathway activity, whole-genome somatic copy number alterations, and mutation frequencies.
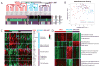
Results: We identified two distinct HPV(+) subtypes (namely HPV-KRT and HPV-IMU). HPV-KRT is characterized by elevated expression of genes in keratinocyte differentiation and oxidation-reduction process, whereas HPV-IMU has strong immune response and mesenchymal differentiation. The differences in expression are likely connected to the differences in HPV characteristics and genomic changes. HPV-KRT has more genic viral integration, lower E2/E4/E5 expression levels, and higher ratio of spliced to full-length HPV oncogene E6 than HPV-IMU; the subgroups also show differences in copy number alterations and mutations, in particular the loss of chr16q in HPV-IMU and gain of chr3q and PIK3CA mutation in HPV-KRT.
Conclusions: Our characterization of two subtypes of HPV(+) HNC tumors provides valuable molecular level information that point to two main carcinogenic paths. Together, these results shed light on stratifications of the HPV(+) HNCs and will help to guide personalized care for HPV(+) HNC patients. Clin Cancer Res; 22(18); 4735-45. ©2016 AACR.
©2016 American Association for Cancer Research.
Conflict of interest statement
The authors declare there are no potential conflicts of interest.
Figures


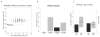

Similar articles
-
Immune Signature-Based Subtypes of Cervical Squamous Cell Carcinoma Tightly Associated with Human Papillomavirus Type 16 Expression, Molecular Features, and Clinical Outcome.Neoplasia. 2019 Jun;21(6):591-601. doi: 10.1016/j.neo.2019.04.003. Epub 2019 May 2. Neoplasia. 2019. PMID: 31055200 Free PMC article.
-
Integrative and comparative genomic analysis of HPV-positive and HPV-negative head and neck squamous cell carcinomas.Clin Cancer Res. 2015 Feb 1;21(3):632-41. doi: 10.1158/1078-0432.CCR-13-3310. Epub 2014 Jul 23. Clin Cancer Res. 2015. PMID: 25056374 Free PMC article.
-
Somatic Host Cell Alterations in HPV Carcinogenesis.Viruses. 2017 Aug 3;9(8):206. doi: 10.3390/v9080206. Viruses. 2017. PMID: 28771191 Free PMC article. Review.
-
Fundamental differences in cell cycle deregulation in human papillomavirus-positive and human papillomavirus-negative head/neck and cervical cancers.Cancer Res. 2007 May 15;67(10):4605-19. doi: 10.1158/0008-5472.CAN-06-3619. Cancer Res. 2007. PMID: 17510386 Free PMC article.
-
Epidemiology of head and neck cancer.Surg Oncol Clin N Am. 2015 Jul;24(3):379-96. doi: 10.1016/j.soc.2015.03.001. Epub 2015 Apr 9. Surg Oncol Clin N Am. 2015. PMID: 25979389 Review.
Cited by
-
Transcriptome and microbiome-immune changes across preinvasive and invasive anal cancer lesions.JCI Insight. 2024 Jul 18;9(16):e180907. doi: 10.1172/jci.insight.180907. JCI Insight. 2024. PMID: 39024554 Free PMC article.
-
Special Issue about Head and Neck Cancers: HPV Positive Cancers.Int J Mol Sci. 2020 May 11;21(9):3388. doi: 10.3390/ijms21093388. Int J Mol Sci. 2020. PMID: 32403287 Free PMC article. Review.
-
Clinical Validity of a Prognostic Gene Expression Cluster-Based Model in Human Papillomavirus-Positive Oropharyngeal Carcinoma.JCO Precis Oncol. 2021 Oct 27;5:PO.21.00094. doi: 10.1200/PO.21.00094. eCollection 2021. JCO Precis Oncol. 2021. PMID: 34738049 Free PMC article.
-
Navigating therapeutic strategies: HPV classification in head and neck cancer.Br J Cancer. 2024 Jul;131(2):220-230. doi: 10.1038/s41416-024-02655-1. Epub 2024 Apr 20. Br J Cancer. 2024. PMID: 38643337 Free PMC article. Review.
-
End of treatment cone-beam computed tomography (CBCT) is predictive of radiation response and overall survival in oropharyngeal squamous cell carcinoma.Radiat Oncol. 2021 Aug 9;16(1):147. doi: 10.1186/s13014-021-01871-w. Radiat Oncol. 2021. PMID: 34372887 Free PMC article.
References
-
- Ferlay J, Shin H-R, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–917. - PubMed
-
- Kreimer AR, Clifford GM, Boyle P, Franceschi S. Human papillomavirus types in head and neck squamous cell carcinomas worldwide: a systematic review. Cancer Epidemiol Biomarkers Prev. 2005;14:467–75. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

