Nanostructured photoelectrochemical solar cell for nitrogen reduction using plasmon-enhanced black silicon
- PMID: 27093916
- PMCID: PMC4842983
- DOI: 10.1038/ncomms11335
Nanostructured photoelectrochemical solar cell for nitrogen reduction using plasmon-enhanced black silicon
Abstract
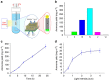
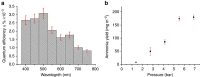
Ammonia (NH3) is one of the most widely produced chemicals worldwide. It has application in the production of many important chemicals, particularly fertilizers. It is also, potentially, an important energy storage intermediate and clean energy carrier. Ammonia production, however, mostly uses fossil fuels and currently accounts for more than 1.6% of global CO2 emissions (0.57 Gt in 2015). Here we describe a solar-driven nanostructured photoelectrochemical cell based on plasmon-enhanced black silicon for the conversion of atmospheric N2 to ammonia producing yields of 13.3 mg m(-2) h(-1) under 2 suns illumination. The yield increases with pressure; the highest observed in this work was 60 mg m(-2) h(-1) at 7 atm. In the presence of sulfite as a reactant, the process also offers a direct solar energy route to ammonium sulfate, a fertilizer of economic importance. Although the yields are currently not sufficient for practical application, there is much scope for improvement in the active materials in this cell.
Figures

Similar articles
-
Recent Advances in Solar Thermal Electrochemical Process (STEP) for Carbon Neutral Products and High Value Nanocarbons.Acc Chem Res. 2019 Nov 19;52(11):3177-3187. doi: 10.1021/acs.accounts.9b00405. Epub 2019 Nov 7. Acc Chem Res. 2019. PMID: 31697061
-
Nitrogen Photoelectrochemical Reduction on TiB2 Surface Plasmon Coupling Allows Us to Reach Enhanced Efficiency of Ammonia Production.ACS Catal. 2023 Aug 3;13(16):10916-10926. doi: 10.1021/acscatal.3c03210. eCollection 2023 Aug 18. ACS Catal. 2023. PMID: 37614521 Free PMC article.
-
Highly Efficient Photoelectrochemical Synthesis of Ammonia Using Plasmon-Enhanced Black Silicon under Ambient Conditions.ACS Appl Mater Interfaces. 2020 May 6;12(18):20376-20382. doi: 10.1021/acsami.0c00881. Epub 2020 Apr 21. ACS Appl Mater Interfaces. 2020. PMID: 32271001
-
Solar Electricity and Solar Fuels: Status and Perspectives in the Context of the Energy Transition.Chemistry. 2016 Jan 4;22(1):32-57. doi: 10.1002/chem.201503580. Epub 2015 Nov 20. Chemistry. 2016. PMID: 26584653 Review.
-
Two-dimensional-related catalytic materials for solar-driven conversion of COx into valuable chemical feedstocks.Chem Soc Rev. 2019 Apr 1;48(7):1972-2010. doi: 10.1039/c8cs00607e. Chem Soc Rev. 2019. PMID: 30357195 Review.
Cited by
-
Scalable Hybrid Antibacterial Surfaces: TiO2 Nanoparticles with Black Silicon.ACS Omega. 2022 Feb 25;7(9):7816-7824. doi: 10.1021/acsomega.1c06706. eCollection 2022 Mar 8. ACS Omega. 2022. PMID: 35284710 Free PMC article.
-
A Rearrangement Reaction to Yield a NH4 + Ion Driven by Polyoxometalate Formation.ACS Omega. 2022 Aug 23;7(35):31474-31481. doi: 10.1021/acsomega.2c04015. eCollection 2022 Sep 6. ACS Omega. 2022. PMID: 36092612 Free PMC article.
-
Design Refinement of Catalytic System for Scale-Up Mild Nitrogen Photo-Fixation.Nanomicro Lett. 2025 Mar 12;17(1):182. doi: 10.1007/s40820-025-01695-3. Nanomicro Lett. 2025. PMID: 40072724 Free PMC article. Review.
-
Paired Chemical Upgrading in Photoelectrochemical Cells.JACS Au. 2025 Apr 23;5(5):2061-2075. doi: 10.1021/jacsau.5c00115. eCollection 2025 May 26. JACS Au. 2025. PMID: 40443900 Free PMC article. Review.
-
Enhanced charge-carrier dynamics and efficient solar-to-urea conversion on Si-based photocathodes.Proc Natl Acad Sci U S A. 2024 Feb 20;121(8):e2311326121. doi: 10.1073/pnas.2311326121. Epub 2024 Feb 13. Proc Natl Acad Sci U S A. 2024. PMID: 38349884 Free PMC article.
References
-
- Jackson R. B. et al.. Reach peak emissions. Nat. Clim. Change 6, 7–10 (2016).
-
- Service R. Chemistry. new recipe produces ammonia from air, water, and sunlight. Science 345, 610 (2014). - PubMed
-
- Ceresana. Market Study: Ammonia (UC-3705) (2012).
-
- Lan R. & Tao S. Ammonia as a suitable fuel for fuel cells. Fuel Cells 2, 35 (2014).
-
- Van Tamelen E. E. & Akermark B. Electrolytic reduction of molecular nitrogen. J. Am. Chem. Soc. 90, 4492–4493 (1968).
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

