Zebrafish Müller glia-derived progenitors are multipotent, exhibit proliferative biases and regenerate excess neurons
- PMID: 27094545
- PMCID: PMC4837407
- DOI: 10.1038/srep24851
Zebrafish Müller glia-derived progenitors are multipotent, exhibit proliferative biases and regenerate excess neurons
Abstract
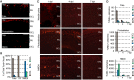
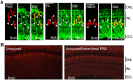
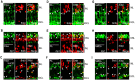
Unlike mammals, zebrafish can regenerate a damaged retina. Key to this regenerative response are Müller glia (MG) that respond to injury by reprogramming and adopting retinal stem cell properties. These reprogrammed MG divide to produce a proliferating population of retinal progenitors that migrate to areas of retinal damage and regenerate lost neurons. Previous studies have suggested that MG-derived progenitors may be biased to produce that are lost with injury. Here we investigated MG multipotency using injury paradigms that target different retinal nuclear layers for cell ablation. Our data indicate that regardless of which nuclear layer was damaged, MG respond by generating multipotent progenitors that migrate to all nuclear layers and differentiate into layer-specific cell types, suggesting that MG-derived progenitors in the injured retina are intrinsically multipotent. However, our analysis of progenitor proliferation reveals a proliferative advantage in nuclear layers where neurons were ablated. This suggests that feedback inhibition from surviving neurons may skew neuronal regeneration towards ablated cell types.
Figures



References
-
- Hitchcock P. F. & Raymond P. A. The teleost retina as a model for developmental and regeneration biology. Zebrafish 1, 257–271 (2004). - PubMed
-
- Otteson D. C. & Hitchcock P. F. Stem cells in the teleost retina: persistent neurogenesis and injury-induced regeneration. Vision Res 43, 927–936 (2003). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

