Essential role for a novel population of binucleated mammary epithelial cells in lactation
- PMID: 27102712
- PMCID: PMC4844753
- DOI: 10.1038/ncomms11400
Essential role for a novel population of binucleated mammary epithelial cells in lactation
Abstract
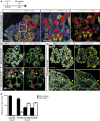
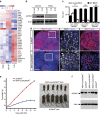
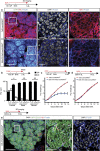
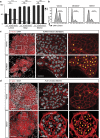
The mammary gland represents a unique tissue to study organogenesis as it predominantly develops in the post-natal animal and undergoes dramatic morphogenetic changes during puberty and the reproductive cycle. The physiological function of the mammary gland is to produce milk to sustain the newborn. Here we view the lactating gland through three-dimensional confocal imaging of intact tissue. We observed that the majority of secretory alveolar cells are binucleated. These cells first arise in very late pregnancy due to failure of cytokinesis and are larger than mononucleated cells. Augmented expression of Aurora kinase-A and Polo-like kinase-1 at the lactogenic switch likely mediates the formation of binucleated cells. Our findings demonstrate an important physiological role for polyploid mammary epithelial cells in lactation, and based on their presence in five different species, suggest that binucleated cells evolved to maximize milk production and promote the survival of offspring across all mammalian species.
Figures


Similar articles
-
Binuclear Cells in the Lactating Mammary Gland: New Insights on an Old Concept?J Mammary Gland Biol Neoplasia. 2016 Jun;21(1-2):21-3. doi: 10.1007/s10911-016-9356-5. Epub 2016 Jun 2. J Mammary Gland Biol Neoplasia. 2016. PMID: 27255141 Review.
-
Inactivation of VEGF in mammary gland epithelium severely compromises mammary gland development and function.FASEB J. 2007 Dec;21(14):3994-4004. doi: 10.1096/fj.07-8720com. Epub 2007 Jul 11. FASEB J. 2007. PMID: 17625068
-
Expression of constitutively activated Akt in the mammary gland leads to excess lipid synthesis during pregnancy and lactation.J Lipid Res. 2003 Jun;44(6):1100-12. doi: 10.1194/jlr.M300045-JLR200. Epub 2003 Apr 16. J Lipid Res. 2003. PMID: 12700340
-
Overexpression of human Cripto-1 in transgenic mice delays mammary gland development and differentiation and induces mammary tumorigenesis.Am J Pathol. 2005 Aug;167(2):585-97. doi: 10.1016/S0002-9440(10)63000-3. Am J Pathol. 2005. PMID: 16049342 Free PMC article.
-
The tammar wallaby and fur seal: models to examine local control of lactation.J Dairy Sci. 2007 Jun;90 Suppl 1:E66-75. doi: 10.3168/jds.2006-483. J Dairy Sci. 2007. PMID: 17517753 Review.
Cited by
-
Binuclear Cells in the Lactating Mammary Gland: New Insights on an Old Concept?J Mammary Gland Biol Neoplasia. 2016 Jun;21(1-2):21-3. doi: 10.1007/s10911-016-9356-5. Epub 2016 Jun 2. J Mammary Gland Biol Neoplasia. 2016. PMID: 27255141 Review.
-
Functional consequences of somatic polyploidy in development.Development. 2024 Mar 1;151(5):dev202392. doi: 10.1242/dev.202392. Epub 2024 Feb 28. Development. 2024. PMID: 38415794 Free PMC article. Review.
-
Reducing the aneuploid cell burden - cell competition and the ribosome connection.Dis Model Mech. 2022 Nov 1;15(11):dmm049673. doi: 10.1242/dmm.049673. Epub 2022 Nov 29. Dis Model Mech. 2022. PMID: 36444717 Free PMC article. Review.
-
Non-coding transcriptomic profiles in the sheep mammary gland during different lactation periods.Front Vet Sci. 2022 Nov 8;9:983562. doi: 10.3389/fvets.2022.983562. eCollection 2022. Front Vet Sci. 2022. PMID: 36425117 Free PMC article.
-
Tissue-resident ductal macrophages survey the mammary epithelium and facilitate tissue remodelling.Nat Cell Biol. 2020 May;22(5):546-558. doi: 10.1038/s41556-020-0505-0. Epub 2020 Apr 27. Nat Cell Biol. 2020. PMID: 32341550
References
-
- Hadsell D. L. et al. Changes in secretory cell turnover, and mitochondrial oxidative damage in the mouse mammary gland during a single prolonged lactation cycle suggest the possibility of accelerated cellular aging. Exp. Gerontol. 41, 271–281 (2006) . - PubMed
-
- Joshi K., Ellis J. T., Hughes C. M., Monaghan P. & Neville A. M. Cellular proliferation in the rat mammary gland during pregnancy and lactation. Lab. Invest. 54, 52–61 (1986) . - PubMed
-
- Knight C. H. & Peaker M. Mammary cell proliferation in mice during pregnancy and lactation in relation to milk yield. Q. J. Exp. Physiol. 67, 165–177 (1982) . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

