A First Tetraplex Assay for the Simultaneous Quantification of Total α-Synuclein, Tau, β-Amyloid42 and DJ-1 in Human Cerebrospinal Fluid
- PMID: 27116005
- PMCID: PMC4846093
- DOI: 10.1371/journal.pone.0153564
A First Tetraplex Assay for the Simultaneous Quantification of Total α-Synuclein, Tau, β-Amyloid42 and DJ-1 in Human Cerebrospinal Fluid
Abstract
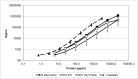
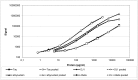
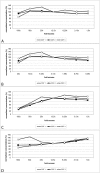
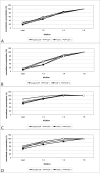
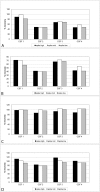
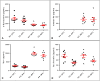
The quantification of four distinct proteins (α-synuclein, β-amyloid1-42, DJ-1, and total tau) in cerebrospinal fluid (CSF) has been proposed as a laboratory-based platform for the diagnosis of Parkinson's disease (PD) and Alzheimer's disease (AD). While there is some clinical utility in measuring these markers individually, their usage in routine clinical testing remains challenging, in part due to substantial overlap of concentrations between healthy controls and diseased subjects. In contrast, measurement of different analytes in a single sample from individual patients in parallel appears to considerably improve the accuracy of AD or PD diagnosis. Here, we report the development and initial characterization of a first, electrochemiluminescence-based multiplex immunoassay for the simultaneous quantification of all four proteins ('tetraplex') in as little as 50 μl of CSF. In analytical performance experiments, we assessed its sensitivity, spike-recovery rate, parallelism and dilution linearity as well as the intra- and inter-assay variability. Using our in-house calibrators, we recorded a lower limit of detection for α-synuclein, β-amyloid42, DJ-1, and t-tau of 1.95, 1.24, 5.63, and 4.05 pg/ml, respectively. The corresponding, linear concentration range covered >3 orders of magnitude. In diluted CSF samples (up to 1:4), spike-recovery rates ranged from a low of 55% for β-amyloid42 to a high of 98% for DJ-1. Hillslopes ranged from 1.03 to 1.30, and inter-assay variability demonstrated very high reproducibility. Our newly established tetraplex assay represents a significant technical advance for fluid-based biomarker studies in neurodegenerative disorders allowing the simultaneous measurement of four pivotal makers in single CSF specimens. It provides exceptional sensitivity, accuracy and speed.
Conflict of interest statement
Figures
Similar articles
-
Simultaneous analysis of cerebrospinal fluid biomarkers using microsphere-based xMAP multiplex technology for early detection of Alzheimer's disease.Methods. 2012 Apr;56(4):484-93. doi: 10.1016/j.ymeth.2012.03.023. Epub 2012 Apr 6. Methods. 2012. PMID: 22503777 Review.
-
Differential role of CSF fatty acid binding protein 3, α-synuclein, and Alzheimer's disease core biomarkers in Lewy body disorders and Alzheimer's dementia.Alzheimers Res Ther. 2017 Jul 28;9(1):52. doi: 10.1186/s13195-017-0276-4. Alzheimers Res Ther. 2017. PMID: 28750675 Free PMC article.
-
Oligomeric α-synuclein and β-amyloid variants as potential biomarkers for Parkinson's and Alzheimer's diseases.Eur J Neurosci. 2016 Jan;43(1):3-16. doi: 10.1111/ejn.13056. Epub 2015 Oct 15. Eur J Neurosci. 2016. PMID: 26332448 Free PMC article.
-
The value of cerebrospinal fluid α-synuclein and the tau/α-synuclein ratio for diagnosis of neurodegenerative disorders with Lewy pathology.Eur J Neurol. 2020 Jan;27(1):43-50. doi: 10.1111/ene.14032. Epub 2019 Jul 24. Eur J Neurol. 2020. PMID: 31293044
-
Discovery, validation and optimization of cerebrospinal fluid biomarkers for use in Parkinson's disease.Expert Rev Mol Diagn. 2017 Aug;17(8):771-780. doi: 10.1080/14737159.2017.1341312. Epub 2017 Jun 19. Expert Rev Mol Diagn. 2017. PMID: 28604235 Review.
Cited by
-
A β-Amyloid(1-42) Biosensor Based on Molecularly Imprinted Poly-Pyrrole for Early Diagnosis of Alzheimer's Disease.J Biomed Phys Eng. 2021 Apr 1;11(2):215-228. doi: 10.31661/jbpe.v0i0.1070. eCollection 2021 Apr. J Biomed Phys Eng. 2021. PMID: 33937128 Free PMC article.
-
Highly sensitive quantification of Alzheimer's disease biomarkers by aptamer-assisted amplification.Theranostics. 2019 May 9;9(10):2939-2949. doi: 10.7150/thno.29232. eCollection 2019. Theranostics. 2019. PMID: 31244934 Free PMC article.
-
Recent Advancements in Electrochemical Biosensors for Alzheimer's Disease Biomarkers Detection.Curr Med Chem. 2021;28(20):4049-4073. doi: 10.2174/0929867327666201111141341. Curr Med Chem. 2021. PMID: 33176635 Free PMC article. Review.
References
-
- Abdi F, Quinn JF, Jankovic J, McIntosh M, Leverenz JB, Peskind E, et al. Detection of biomarkers with a multiplex quantitative proteomic platform in cerebrospinal fluid of patients with neurodegenerative disorders. J Alzheimers Dis. 2006;9: 293–348. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

