Platelet-cytokine Complex Suppresses Tumour Growth by Exploiting Intratumoural Thrombin-dependent Platelet Aggregation
- PMID: 27117228
- PMCID: PMC4846878
- DOI: 10.1038/srep25077
Platelet-cytokine Complex Suppresses Tumour Growth by Exploiting Intratumoural Thrombin-dependent Platelet Aggregation
Abstract
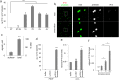
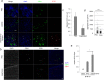
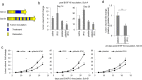
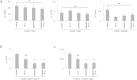
Tumours constitute unique microenvironments where various blood cells and factors are exposed as a result of leaky vasculature. In the present study, we report that thrombin enrichment in B16F10 melanoma led to platelet aggregation, and this property was exploited to administer an anticancer cytokine, interferon-gamma induced protein 10 (IP10), through the formation of a platelet-IP10 complex. When intravenously infused, the complex reached platelet microaggregates in the tumour. The responses induced by the complex were solely immune-mediated, and tumour cytotoxicity was not observed. The complex suppressed the growth of mouse melanoma in vivo, while both platelets and the complex suppressed the accumulation of FoxP3(+) regulatory T cells in the tumour. These results demonstrated that thrombin-dependent platelet aggregation in B16F10 tumours defines platelets as a vector to deliver anticancer cytokines and provide specific treatment benefits.
Figures

Similar articles
-
Expression of a Mr 41,000 glycoprotein associated with thrombin-independent platelet aggregation in high metastatic variants of murine B16 melanoma.Cancer Res. 1990 Oct 15;50(20):6657-62. Cancer Res. 1990. PMID: 2208129
-
Coagulation Factor XI Promotes Distal Platelet Activation and Single Platelet Consumption in the Bloodstream Under Shear Flow.Arterioscler Thromb Vasc Biol. 2016 Mar;36(3):510-7. doi: 10.1161/ATVBAHA.115.307034. Epub 2016 Jan 14. Arterioscler Thromb Vasc Biol. 2016. PMID: 26769048 Free PMC article.
-
Platelet-derived microparticles regulates thrombin generation via phophatidylserine in abdominal sepsis.J Cell Physiol. 2018 Feb;233(2):1051-1060. doi: 10.1002/jcp.25959. Epub 2017 Jun 6. J Cell Physiol. 2018. PMID: 28409836
-
The interaction of thrombin with blood platelets.Platelets. 2005 Nov;16(7):373-85. doi: 10.1080/09537100500123568. Platelets. 2005. PMID: 16236598 Review.
-
Undercover Agents: Targeting Tumours with Modified Platelets.Trends Cancer. 2017 Mar;3(3):235-246. doi: 10.1016/j.trecan.2017.01.006. Epub 2017 Feb 27. Trends Cancer. 2017. PMID: 28718434 Review.
Cited by
-
Platelets and platelet extracellular vesicles in drug delivery therapy: A review of the current status and future prospects.Front Pharmacol. 2022 Oct 18;13:1026386. doi: 10.3389/fphar.2022.1026386. eCollection 2022. Front Pharmacol. 2022. PMID: 36330089 Free PMC article. Review.
-
Proteomics reveals differentially regulated pathways when comparing grade 2 and 4 astrocytomas.PLoS One. 2023 Nov 15;18(11):e0290087. doi: 10.1371/journal.pone.0290087. eCollection 2023. PLoS One. 2023. PMID: 37967105 Free PMC article.
-
Bidirectional Interaction Between Cancer Cells and Platelets Provides Potential Strategies for Cancer Therapies.Front Oncol. 2021 Oct 14;11:764119. doi: 10.3389/fonc.2021.764119. eCollection 2021. Front Oncol. 2021. PMID: 34722319 Free PMC article. Review.
-
Development of cancer immunotherapy based on PD-1/PD-L1 pathway blockade.RSC Adv. 2019 Oct 22;9(58):33903-33911. doi: 10.1039/c9ra04590b. eCollection 2019 Oct 18. RSC Adv. 2019. PMID: 35528929 Free PMC article. Review.
-
Identification of berberine as a direct thrombin inhibitor from traditional Chinese medicine through structural, functional and binding studies.Sci Rep. 2017 Mar 9;7:44040. doi: 10.1038/srep44040. Sci Rep. 2017. PMID: 28276481 Free PMC article.
References
-
- Junttila M. R. & de Sauvage F. J. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature 501, 346–354 (2013). - PubMed
-
- Farnsworth R. H., Lackmann M., Achen M. G. & Stacker S. A. Vascular remodeling in cancer. Oncogene 33, 3496–3505 (2014). - PubMed
-
- Holash J., Wiegand S. J. & Yancopoulos G. D. New model of tumor angiogenesis: dynamic balance between vessel regression and growth mediated by angiopoietins and VEGF. Oncogene 18, 5356–5362 (1999). - PubMed
-
- Wu A. M. & Senter P. D. Arming antibodies: prospects and challenges for immunoconjugates. Nat. Biotechnol. 23, 1137–1146 (2005). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

