Optimal ROS Signaling Is Critical for Nuclear Reprogramming
- PMID: 27117405
- PMCID: PMC4856580
- DOI: 10.1016/j.celrep.2016.03.084
Optimal ROS Signaling Is Critical for Nuclear Reprogramming
Abstract
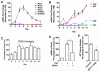
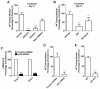
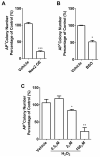
Efficient nuclear reprogramming of somatic cells to pluripotency requires activation of innate immunity. Because innate immune activation triggers reactive oxygen species (ROS) signaling, we sought to determine whether there was a role of ROS signaling in nuclear reprogramming. We examined ROS production during the reprogramming of doxycycline (dox)-inducible mouse embryonic fibroblasts (MEFs) carrying the Yamanaka factors (Oct4, Sox2, Klf4, and c-Myc [OSKM]) into induced pluripotent stem cells (iPSCs). ROS generation was substantially increased with the onset of reprogramming. Depletion of ROS via antioxidants or Nox inhibitors substantially decreased reprogramming efficiency. Similarly, both knockdown and knockout of p22(phox)-a critical subunit of the Nox (1-4) complex-decreased reprogramming efficiency. However, excessive ROS generation using genetic and pharmacological approaches also impaired reprogramming. Overall, our data indicate that ROS signaling is activated early with nuclear reprogramming, and optimal levels of ROS signaling are essential to induce pluripotency.
Keywords: CRISPR/Cas9; NADPH oxidase; Nrf2; iPSCs; nuclear reprogramming; reactive oxygen species.
Copyright © 2016 The Authors. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Retinoic Acid Inducible Gene 1 Protein (RIG1)-Like Receptor Pathway Is Required for Efficient Nuclear Reprogramming.Stem Cells. 2017 May;35(5):1197-1207. doi: 10.1002/stem.2607. Epub 2017 Mar 27. Stem Cells. 2017. PMID: 28276156 Free PMC article.
-
Nuclear S-Nitrosylation Defines an Optimal Zone for Inducing Pluripotency.Circulation. 2019 Sep 24;140(13):1081-1099. doi: 10.1161/CIRCULATIONAHA.119.042371. Epub 2019 Aug 15. Circulation. 2019. PMID: 31412725 Free PMC article.
-
Epithelial cell adhesion molecule (EpCAM) complex proteins promote transcription factor-mediated pluripotency reprogramming.J Biol Chem. 2011 Sep 23;286(38):33520-32. doi: 10.1074/jbc.M111.256164. Epub 2011 Jul 28. J Biol Chem. 2011. PMID: 21799003 Free PMC article.
-
Transflammation: How Innate Immune Activation and Free Radicals Drive Nuclear Reprogramming.Antioxid Redox Signal. 2018 Jul 10;29(2):205-218. doi: 10.1089/ars.2017.7364. Epub 2018 Apr 26. Antioxid Redox Signal. 2018. PMID: 29634341 Free PMC article. Review.
-
Transflammation: Innate immune signaling in nuclear reprogramming.Adv Drug Deliv Rev. 2017 Oct 1;120:133-141. doi: 10.1016/j.addr.2017.09.010. Epub 2017 Sep 13. Adv Drug Deliv Rev. 2017. PMID: 28916494 Free PMC article. Review.
Cited by
-
Hydrogen Peroxide and Redox Regulation of Developments.Antioxidants (Basel). 2018 Nov 6;7(11):159. doi: 10.3390/antiox7110159. Antioxidants (Basel). 2018. PMID: 30404180 Free PMC article. Review.
-
Physiological Signaling Functions of Reactive Oxygen Species in Stem Cells: From Flies to Man.Front Cell Dev Biol. 2021 Aug 6;9:714370. doi: 10.3389/fcell.2021.714370. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34422833 Free PMC article. Review.
-
Vitamin C facilitates direct cardiac reprogramming by inhibiting reactive oxygen species.Stem Cell Res Ther. 2024 Jan 17;15(1):19. doi: 10.1186/s13287-023-03615-x. Stem Cell Res Ther. 2024. PMID: 38229180 Free PMC article.
-
Role of angiogenic transdifferentiation in vascular recovery.Front Cardiovasc Med. 2023 May 2;10:1155835. doi: 10.3389/fcvm.2023.1155835. eCollection 2023. Front Cardiovasc Med. 2023. PMID: 37200975 Free PMC article. Review.
-
Targeting the redox system for cardiovascular regeneration in aging.Aging Cell. 2023 Dec;22(12):e14020. doi: 10.1111/acel.14020. Epub 2023 Nov 13. Aging Cell. 2023. PMID: 37957823 Free PMC article. Review.
References
-
- Anrather J, Racchumi G, Iadecola C. NF-kappaB regulates phagocytic NADPH oxidase by inducing the expression of gp91phox. The Journal of biological chemistry. 2006;281:5657–5667. - PubMed
-
- Armstrong L, Tilgner K, Saretzki G, Atkinson SP, Stojkovic M, Moreno R, Przyborski S, Lako M. Human induced pluripotent stem cell lines show stress defense mechanisms and mitochondrial regulation similar to those of human embryonic stem cells. Stem cells. 2010;28:661–673. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

