Pipelle for Pregnancy (PIP): study protocols for three randomised controlled trials
- PMID: 27121007
- PMCID: PMC4847336
- DOI: 10.1186/s13063-016-1301-9
Pipelle for Pregnancy (PIP): study protocols for three randomised controlled trials
Abstract
Background: The success rate of infertility treatments remains modest. Endometrial injury has been suggested as an intervention to increase the probability of pregnancy in women undergoing assisted reproductive technologies such as in vitro fertilisation (IVF). The majority of studies and systematic reviews have reported that endometrial injury improves the outcomes of IVF, intrauterine insemination and natural conception; however, the size and quality of the studies are poor. The low quality of the available evidence questions the presence of any real beneficial effect, and the applicability of the intervention in different populations remains unclear.
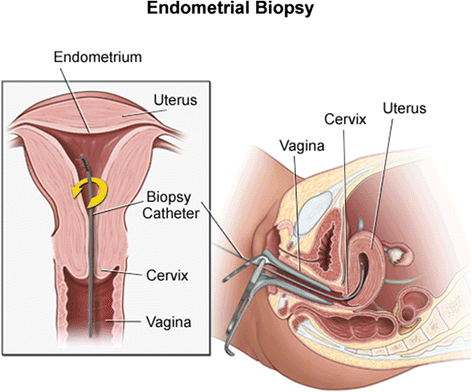
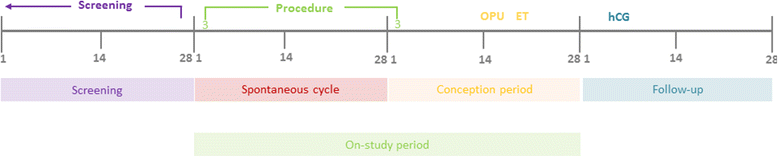
Methods/design: The PIP trials are three multi-centre, randomised controlled trials designed to test three separate hypotheses: whether endometrial injury increases the probability of live birth in women or couples 1) who are undergoing autologous embryo transfer as part of an IVF cycle (PIP-IVF), 2) with unexplained infertility who are attempting to conceive naturally (PIP-UE) and 3) with subfertility related to polycystic ovarian syndrome (PCOS) who are on ovulation induction medication and attempting to conceive (PIP-PCOS). Participants will be randomised to either undergo endometrial injury by endometrial pipelle biopsy or to: • no intervention (PIP-IVF), or • a sham procedure (PIP-PCOS and PIP-UE). In PIP-IVF, endometrial injury will be carried out between day three of the cycle prior to the IVF cycle, and day three of the IVF cycle. In PIP-UE and PIP-PCOS, endometrial injury or a sham procedure will be undertaken between days 1-12 of a menstrual cycle or ovulation induction cycle respectively. Participants in PIP-UE and PIP-PCOS will then be followed for three cycles during which time they will attempt to conceive from sexual intercourse. To ensure allocation concealment, randomisation will be carried out using a web-based system or sequentially numbered, opaque, sealed envelopes. The primary outcome is live birth. Secondary outcomes include ongoing pregnancy, clinical pregnancy and miscarriage. The required sample sizes for the PIP studies have been estimated at 840 (PIP-IVF), 350 (PIP-UE) and 280 (PIP-PCOS). Primary analysis will be as per intention-to-treat principles.
Discussion: The PIP trials are designed to address the gaps in the utility of endometrial scratching as a treatment for subfertility in three different populations. If the beneficial effect of this intervention can be confirmed in these settings, endometrial scratching will provide a cost-effective method for helping women and couples to conceive.
Trial registrations: PIP-IVF ACTRN12614000626662 registered 13/6/2014; PIP-PCOS ACTRN12614000657628 registered 24/6/2014; PIP-UE ACTRN12614000656639 registered 24/6/2014. The trials are ongoing.
Keywords: Endometrial injury; Endometrial receptivity; Endometrial scratching; Implantation; Inflammation.
Figures

References
-
- Macaldowie A, Wang Y, Chambers G, Sullivan E. Assisted reproductive technology in Australia and New Zealand 2011. UNSW 2013. https://npesu.unsw.edu.au/sites/default/files/npesu/data_collection/Assi....
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

