Hypoxia promotes Rab5 activation, leading to tumor cell migration, invasion and metastasis
- PMID: 27121131
- PMCID: PMC5045416
- DOI: 10.18632/oncotarget.8794
Hypoxia promotes Rab5 activation, leading to tumor cell migration, invasion and metastasis
Abstract
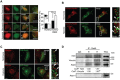
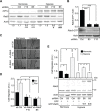
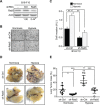
Hypoxia, a common condition of the tumor microenvironment, is associated with poor patient prognosis, tumor cell migration, invasion and metastasis. Recent evidence suggests that hypoxia alters endosome dynamics in tumor cells, leading to augmented cell proliferation and migration and this is particularly relevant, because endosomal components have been shown to be deregulated in cancer. The early endosome protein Rab5 is a small GTPase that promotes integrin trafficking, focal adhesion turnover, Rac1 activation, tumor cell migration and invasion. However, the role of Rab5 and downstream events in hypoxia remain unknown. Here, we identify Rab5 as a critical player in hypoxia-driven tumor cell migration, invasion and metastasis. Exposure of A549 human lung carcinoma, ZR-75, MDA-MB-231 and MCF-7 human breast cancer and B16-F10 mouse melanoma cells to hypoxia increased Rab5 activation, followed by its re-localization to the leading edge and association with focal adhesions. Importantly, Rab5 was required for hypoxia-driven cell migration, FAK phosphorylation and Rac1 activation, as shown by shRNA-targeting and transfection assays with Rab5 mutants. Intriguingly, the effect of hypoxia on both Rab5 activity and migration was substantially higher in metastatic B16-F10 cells than in poorly invasive B16-F0 cells. Furthermore, exogenous expression of Rab5 in B16-F0 cells predisposed to hypoxia-induced migration, whereas expression of the inactive mutant Rab5/S34N prevented the migration of B16-F10 cells induced by hypoxia. Finally, using an in vivo syngenic C57BL/6 mouse model, Rab5 expression was shown to be required for hypoxia-induced metastasis. In summary, these findings identify Rab5 as a key mediator of hypoxia-induced tumor cell migration, invasion and metastasis.
Keywords: Rab5; hypoxia; metastasis; migration; tumor.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Rab5 activation promotes focal adhesion disassembly, migration and invasiveness in tumor cells.J Cell Sci. 2013 Sep 1;126(Pt 17):3835-47. doi: 10.1242/jcs.119727. Epub 2013 Jun 26. J Cell Sci. 2013. PMID: 23813952 Free PMC article.
-
Rab5 is required in metastatic cancer cells for Caveolin-1-enhanced Rac1 activation, migration and invasion.J Cell Sci. 2014 Jun 1;127(Pt 11):2401-6. doi: 10.1242/jcs.141689. Epub 2014 Mar 21. J Cell Sci. 2014. PMID: 24659799 Free PMC article.
-
Focal adhesion kinase-dependent activation of the early endocytic protein Rab5 is associated with cell migration.J Biol Chem. 2019 Aug 23;294(34):12836-12845. doi: 10.1074/jbc.RA119.008667. Epub 2019 Jul 10. J Biol Chem. 2019. PMID: 31292193 Free PMC article.
-
On the role of Rab5 in cell migration.Curr Mol Med. 2014 Feb;14(2):235-45. doi: 10.2174/1566524014666140128111347. Curr Mol Med. 2014. PMID: 24467205 Review.
-
Rab5 in the regulation of cell motility and invasion.Curr Protein Pept Sci. 2011 Feb;12(1):43-51. doi: 10.2174/138920311795659461. Curr Protein Pept Sci. 2011. PMID: 21190523 Review.
Cited by
-
Exosomes: a promising avenue for cancer diagnosis beyond treatment.Front Cell Dev Biol. 2024 Feb 13;12:1344705. doi: 10.3389/fcell.2024.1344705. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38419843 Free PMC article. Review.
-
A new role for human dyskerin in vesicular trafficking.FEBS Open Bio. 2017 Sep 12;7(10):1453-1468. doi: 10.1002/2211-5463.12307. eCollection 2017 Oct. FEBS Open Bio. 2017. PMID: 28979836 Free PMC article.
-
Comprehensive Analysis of Expression, Clinicopathological Association and Potential Prognostic Significance of RABs in Pancreatic Cancer.Int J Mol Sci. 2020 Aug 4;21(15):5580. doi: 10.3390/ijms21155580. Int J Mol Sci. 2020. PMID: 32759795 Free PMC article.
-
Silencing Rab14 represses the proliferation and migration of oral squamous cell carcinoma, and enhances cisplatin sensitivity.Am J Transl Res. 2017 Sep 15;9(9):4195-4205. eCollection 2017. Am J Transl Res. 2017. PMID: 28979693 Free PMC article.
-
Association of RAB5 overexpression in pancreatic cancer with cancer progression and poor prognosis via E-cadherin suppression.Oncotarget. 2017 Feb 14;8(7):12290-12300. doi: 10.18632/oncotarget.14703. Oncotarget. 2017. PMID: 28103577 Free PMC article.
References
-
- Brahimi-Horn MC, Chiche J, Pouyssegur J. Hypoxia and cancer. Journal of molecular medicine. 2007;85:1301–1307. - PubMed
-
- Vaupel P. Hypoxia and aggressive tumor phenotype: implications for therapy and prognosis. The oncologist. 2008;13(Suppl 3):21–26. - PubMed
-
- Vaupel P, Mayer A. Hypoxia in cancer: significance and impact on clinical outcome. Cancer metastasis reviews. 2007;26:225–239. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

